GTPase-powered progressive contraction of a supramolecular ring driving chloroplast division
GTPase-powered progressive contraction of a supramolecular ring driving chloroplast division
Yoshida, Y.; Mogi, Y.; Kuroiwa, H.; Kuroiwa, T.
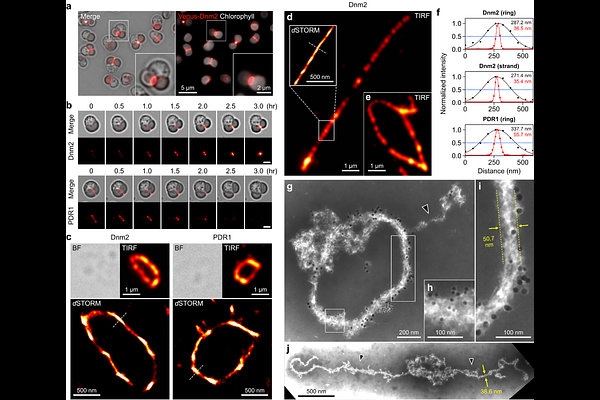
AbstractThe chloroplast division machinery, known as the division ring, is a supramolecular complex composed of bacterial- and host-derived proteins. However, how the division ring generates the force required to sever a chloroplast remains poorly understood. Here, we established an in vitro assay in which chloroplasts isolated from Cyanidioschyzon merolae undergo GTP hydrolysis-dependent division. Using this assay, we show that Dnm2, rather than FtsZ, acts as the motor driving GTPase-powered contraction of the division ring, thereby physically dividing the chloroplast. We further demonstrate that the division ring is assembled through coiling of a glycosyltransferase-mediated filament and is crosslinked by dimerization of the Dnm2 GTPase domain. Following GTP hydrolysis-dependent force generation, Dnm2 retains its dimeric form in GDP-bound and nucleotide-free states, providing a locking step that suppresses back-slippage of the coiling ring during division. Thus, this mechanical design enables progressive, ratchet-like constriction of the division ring through coiling, overcoming the mechanical load posed by the chloroplast and generating the force required for fission, consistent with quantitative simulations. These findings suggest that a specialized division-ring mechanism, distinct from vesicle fission systems, evolved to mediate endosymbiont fission, allowing host control of endosymbiont proliferation and promoting faithful inheritance of the emerging organelle.