EspH utilizes phosphoinositide and Rab binding domains to interact with plasma membrane infection sites and Rab GTPases
EspH utilizes phosphoinositide and Rab binding domains to interact with plasma membrane infection sites and Rab GTPases
Aroeti, B.; Nandi, I.; Ramachandran, R. P.; Shalev, D. E.; Schneidman-Duhovny, D.; Rahav, R. S.; Melamed-Book, N.; Rosenshine, I.
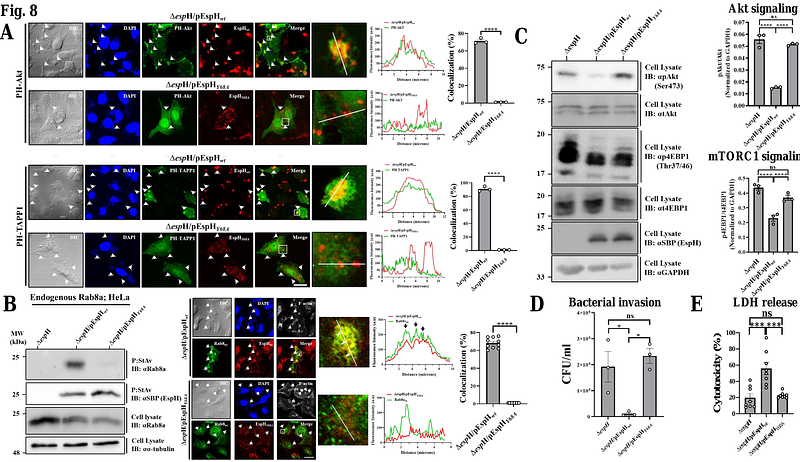
AbstractEnteropathogenic E. coli (EPEC) is a Gram-negative bacterial pathogen that causes persistent diarrhea. Upon attachment to the apical plasma membrane of the intestinal epithelium, the pathogen translocates virulent proteins called effectors into the infected cells. These effectors hijack numerous host processes for the pathogen\'s benefit. Therefore, studying the mechanisms underlying their action is crucial for a better understanding of the disease. We show that translocated EspH interacts with multiple host Rab GTPases. AlphaFold predictions and site-directed mutagenesis identified glutamic acid and lysine at positions 37 and 41 as Rab interacting residues. Mutating these sites abolished the EspH ability to inhibit the Akt and mTORC1 signaling, lysosomal exocytosis, and bacterial invasion. Knocking out the endogenous Rab8a gene expression highlighted the involvement of Rab8a in Akt/mTORC1 signaling and lysosomal exocytosis. A phosphoinositide binding domain with a critical tyrosine was identified in EspH. Mutating the tyrosine abolished the localization of EspH at infection sites and its capacity to interact with Rabs. Our data suggest novel EspH-dependent mechanisms that elicit immune signaling and membrane trafficking during EPEC infection.


