Metabolic and Thermal Cues Shape IL 6 Responses and Disease Tolerance Mechanisms in Severe Malaria
Metabolic and Thermal Cues Shape IL 6 Responses and Disease Tolerance Mechanisms in Severe Malaria
ANAGU, L. O.; Wassmer, S. C.; Anagboso, I.; Elo-ilo, J.; Ezeagwuna, D.; Amambua-Ngwa, A. C.
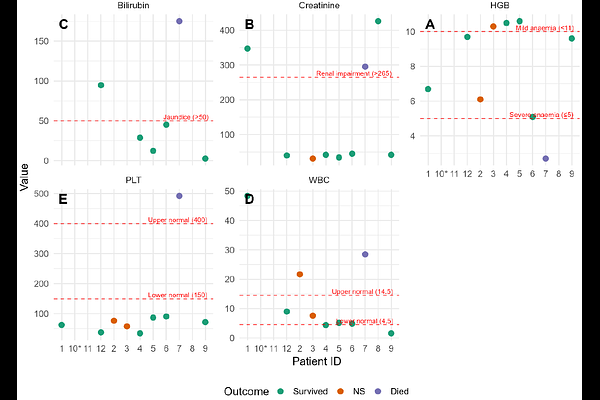
AbstractSevere malaria disproportionately affects children during their earliest Plasmodium falciparum infections, when immunopathology rather than parasite burden often drives clinical deterioration. Because direct investigation of host-parasite interactions during severe disease is ethically impossible, we developed a two-dimensional ex vivo co-culture system that recapitulates key physiological features of malaria pathogenesis. Using PBMCs from malaria-naive and malaria-exposed adults co-cultured with a freshly adapted P. falciparum isolate, we modelled the combined effects of febrile temperature, pipecolic acid (PA), and lysophosphatidylcholine (LPC) depletion on IL-6 secretion. We also integrated clinical data from children with severe malaria in Anambra State, Nigeria. Across the conditions, IL-6 output was not driven by temperature alone but by a metabolically gated interaction: febrile temperature amplified IL-6 only when PA was present, and LPC was not limiting. LPC depletion suppressed IL-6 to near-baseline levels regardless of temperature or PA, indicating that lipid availability constrains inflammatory signalling. Clinical data showed that adverse outcomes clustered with markers of multi-organ dysfunction. Together, these findings support a model in which IL-6 is a context-dependent mediator - participating in inflammatory pathways but not acting as a singular causal driver - and in which metabolic stress, febrile cues, and host tolerance mechanisms jointly shape cytokine production. Ongoing bioinformatics analysis will define the transcriptional responses of both parasite and host cells under these malaria-relevant conditions.