Functional Divergence and Conservation in the QueC Protein Family (PF06508): From tRNA Modification to Anti-Phage Defense
Functional Divergence and Conservation in the QueC Protein Family (PF06508): From tRNA Modification to Anti-Phage Defense
Libby, K. M.; Benda, R.; de Crecy-Lagard, V.; Hutinet, G.
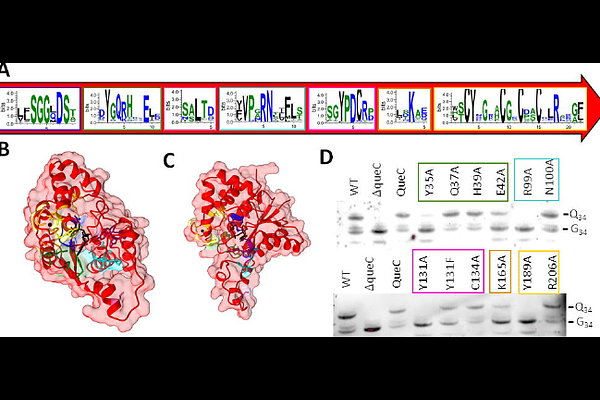
AbstractThe QueC protein family (PF06508) is best known for its role in the biosynthesis of 7-deazaguanine derivatives, including the tRNA modification queuosine and the DNA base 7-cyano-7-deazaguanine (preQ0). Recent discoveries, however, reveal that this ancient scaffold has been repeatedly repurposed for distinct biological functions, including roles in anti-phage defense. Here, we combine Sequence Similarity Networks (SSN) with genomic neighborhood analyses to map the functional diversification of the PF06508 superfamily. We delineate the canonical QueC cluster and experimentally define a refined catalytic signature, identifying previously unrecognized residues essential for tRNA modification. Beyond the canonical function, we characterize the evolutionary repurposing of the QueC fold in anti-phage systems, distinguishing between the "divergent specialist" QatC, which remodels the active site, and the "minimalist conservationist" Cap9, which retains the ancestral catalytic core. We further uncover the expansion of the superfamily into additional biochemical pathways, including cofactor biosynthesis (e.g., LarE) and purine salvage. Together, these findings provide a comprehensive framework for understanding how the QueC scaffold has been adapted from small-molecule biosynthesis to roles in protein modification and phage defense.