Sequence and structure of protein binding sites in RNA impact biomolecular condensates
Sequence and structure of protein binding sites in RNA impact biomolecular condensates
Cole, S. J.; Allen, S. R.; Guzman, B. B.; Hu, Y.; Stormo, B. M.; Roden, C. A.; Ekena, J.; Zhang, V.; McLaughlin, G. A.; Crocker, A. W.; Laederach, A.; Dominguez, D.; Gladfelter, A. S.
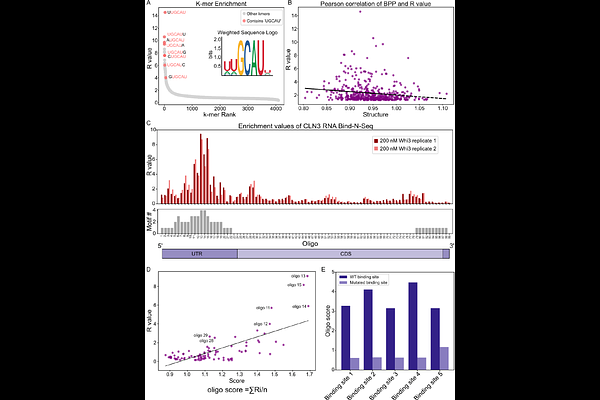
AbstractBiomolecular condensates are central to subcellular compartmentalization. Although many condensates contain and regulate RNA, research has primarily focused on protein interactions. Here, we investigate RNA-protein interactions underlying cell cycle-regulating condensates in the multinucleate fungus Ashbya gossypii. These condensates form through interactions between G1 cyclin mRNA CLN3 and RNA-binding protein Whi3, which was predicted by homology to recognize a five-nucleotide motif repeated within the transcript. Natural variation in motif number in Ashbya strains led us to hypothesize that binding site valence may influence condensate properties. Using unbiased binding assays, we determined the preference of Ashbya Whi3 protein for specific primary RNA sequence and mutated individual Whi3-binding sites within CLN3 mRNA. Mutants exhibited distinct condensate properties despite having the same valence in terms of binding site number. Mutations altered the saturation concentration (Csat) and dense phase concentration of RNA and protein in cell-free reconstitution experiments. A subset of mutants, showed reduced number of condensates and deregulation of the cell cycle in cells. We also find that enhanced availability of single-stranded RNA can compensate for loss of binding sites. Together, these data indicate that differences in RNA protein binding-site context and not simply valence plays a critical role in determining condensate properties.