Within-host population structure, migration, and parallel adaptive evolution of Pseudomonas aeruginosa in cystic fibrosis lung disease
Within-host population structure, migration, and parallel adaptive evolution of Pseudomonas aeruginosa in cystic fibrosis lung disease
Ritz, D.; Clay, M. E.; Kim, T.; Van Gelder, R. D.; Chandrashekhar, J. H.; Collins, A. J.; Goddard, J.; Ashare, A.; Hoehn, K. B.; Schultz, D.; Whitaker, R. J.; Hogan, D. A.
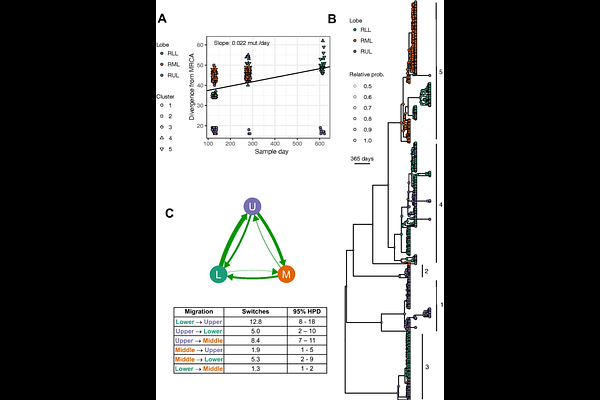
AbstractPseudomonas aeruginosa infections in adults with cystic fibrosis (CF) are comprised of heterogeneous populations, most often tracing ancestry back to a single recent common ancestor. What is not clear is the physical spatial structure within the lung infection population, its stability over time and whether this physical structure leads to different evolutionary trajectories in different adaptive environments. To compare the P. aeruginosa populations across a single lung, we performed whole genome sequence analyses of 450 isolates recovered from lavage samples of the three different lobes of the right lung from a person with mild-to-moderate CF lung disease at three time points over the course of ~1.5 years. We found that isolates fell into five distinct phylogenetic lineages with evidence for repeated translocation of isolates from different lineages across lobes and loss-of-function mutations in lasR and mucA were present in all 450 isolates. The well-resolved phylogenetic analyses revealed a structured population in which we find the coexistence of a slowly evolving lineage and more rapidly evolving lineages. There is also support for numerous migration events. Further, strong evidence for parallel adaptive mutations in multiple genes revealed distinct evolutionary paths affecting mucoid phenotypes and genetic variation in antibiotic resistance-associated pathways across coexisting populations within a single individual over time. These results provide an example of within-host evolution leading to microheterogeneity that may be useful to consider in future study of infection metapopulations dynamics over the course of chronic infection.