Proton tunneling at the ryanodine receptor Ca2+ activation site provides temperature-invariant noise for robust Ca2+-induced Ca2+ release
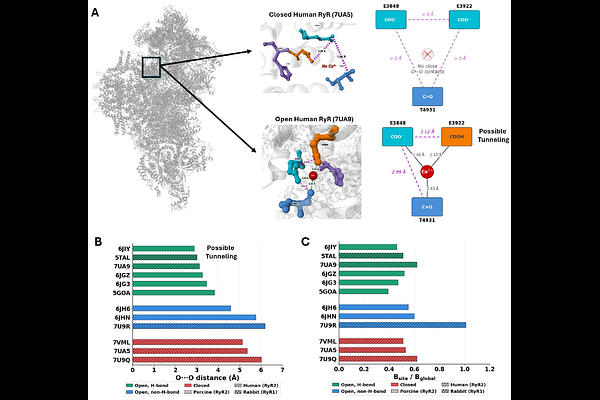
Proton tunneling at the ryanodine receptor Ca2+ activation site provides temperature-invariant noise for robust Ca2+-induced Ca2+ release
Maltsev, A. V.; Lakatta, E. G.; Maltsev, V. A.
AbstractRyanodine receptor (RyR)-mediated calcium-induced calcium release (CICR) is a regenerative trigger-to-release mechanism used in diverse cell types. RyRs amplify small calcium signals into large, localized surges that drive autonomous oscillation, excitation-contraction coupling, pulsatile secretion, neurotransmitter release, or memory formation. Robust RyR function depends on the probability of RyR recruitment remaining within an operating range as physiological conditions change. This raises a basic question: what stabilizes the microscopic calcium-sensitive opening step of RyR across temperature and cellular state? Here we tested that proton tunneling at the conserved RyR2 calcium activation site contributes a temperature-stable stochastic component to channel opening. To this end we developed a multiscale quantum-structural pipeline that estimates the per-channel modulation amplitude from cryo-EM geometry under stated assumptions. We applied this pipeline to a rabbit sinoatrial node cell model with explicit calcium release unit architecture. Holding this RyR quantum noise constant across 25-37{degrees}C preserves pacemaker rhythmicity at low temperature, whereas a Q10-scaled classical noise results in irregular pacemaking activity as amplitude falls below the coherence-resonance range. Experimentally in isolated rabbit sinoatrial node cells, calcium transient rate increased with temperature, but inter-transient interval variability did not, consistent with our numerical model predictions. These results support a model in which proton tunneling stabilizes the regenerative RyR-mediated CICR step. In autonomous oscillators, such as sinoatrial node cells, this manifests as robust rhythmicity. In triggered systems, it is expected to stabilize CICR gain, release synchrony, and release fidelity. The importance of this shared microscopic mechanism is evidenced by the conservation of the calcium-activation site across RyR isoforms for >600 million years of evolution.