Modeling disorder, secondary structure formation, and amyloid growth in FG-nucleoporins
Modeling disorder, secondary structure formation, and amyloid growth in FG-nucleoporins
Dekker, M.; Chen, S. M. H.; Adupa, V.; Onck, P. R.
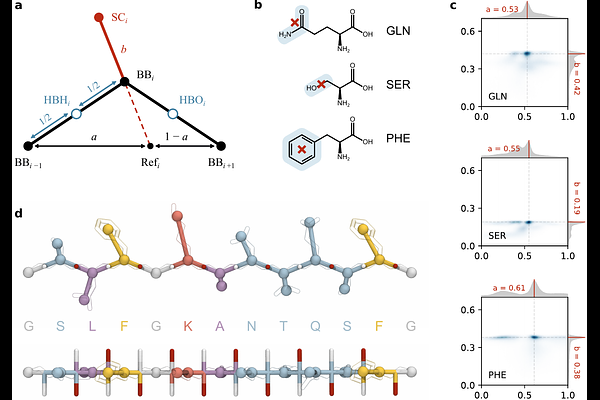
AbstractNuclear transport relies on intrinsically disordered FG-nucleoporins (FG-Nups) that form a dynamic selective barrier, yet experiments show that these proteins can also adopt highly ordered fibrillar structures. Capturing this duality within a single computational framework remains a major challenge. Here, we introduce 2BPA-HB, a sequence-resolved coarse-grained model that combines explicit, directional backbone hydrogen bonding with residue-specific side chain interaction centers derived from all-atom simulations. This design enables microsecond-scale simulations of the transitions between disordered, condensed, and amyloid-like states while maintaining computational efficiency. We first validate the model by simulating several experimentally resolved Nup98 fibril polymorphs, which retain their characteristic {beta}-sheet registry and protofibril architecture. Seeded growth simulations further show that disordered FG fragments align with and extend these fibrillar templates in a sequence-specific manner, and can initiate secondary nucleation events on fibril surfaces. Applying the model to yeast FG-Nups, we find that cohesive FG domains form stable condensates with interaction networks dominated by FG motif contacts, consistent with experimental observations and prior simulations. The increased structural resolution of 2BPA-HB additionally reveals transient - and {beta}-structures within these condensates. Together, these results demonstrate that 2BPA-HB captures both liquid-like and fibrillar behavior of intrinsically disordered proteins within a single coarse-grained framework. The model provides a platform to study how disorder, secondary structure formation, and aggregation coexist in nuclear pore biology, and represents a step forward to a transferable coarse-grained approach for low-complexity proteins that undergo both phase separation and amyloid formation, as implicated in neurodegenerative diseases.