SIRPA suppresses integrin-dependent virus endocytosis
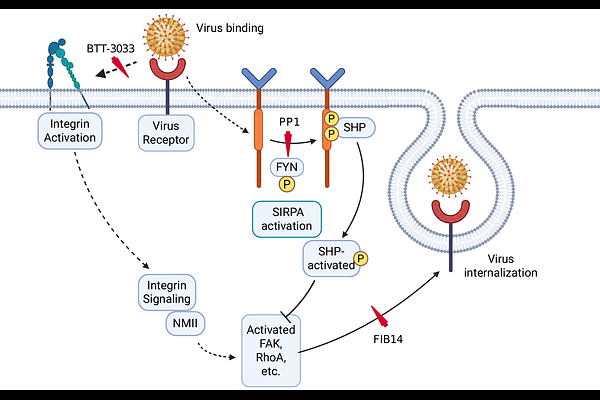
SIRPA suppresses integrin-dependent virus endocytosis
Yan, Z.; Iyer, K.; Li, M.; Jung, K. H.; Hu, C. T.; Ansin, N.; Sarute, N.; Gowen, B. B.; Ross, S. R.
AbstractNew World arenaviruses (NWAs) that cause viral hemorrhagic fever, such as Junin virus, have few therapeutic options. Entry of these viruses into cells is mediated by binding to cell surface receptors, followed by endocytosis and trafficking to a low pH compartment. We showed previously that Signal Regulatory Protein Alpha (SIRPA), a critical cell surface receptor that inhibits macrophage phagocytic activity, decreases internalization by NWAs as well as other pathogenic RNA viruses that traffic to low pH compartments. Here we demonstrate that proteins involved in the SIRPA/integrin signaling axis, including Src homology region 2 (SH2)-containing protein tyrosine phosphatase 2 (SHP2), src family kinases (SFKs), particularly FYN, focal adhesion kinase (FAK), and alpha-integrin play a role in viral endocytosis and that SIRPA inhibits virus entry through blocking this pathway. In addition to defining a role for integrins in viral entry, these studies also provide additional insight into SIRPA interference in processes dependent on integrin signaling, including phagocytosis. Moreover, using drugs that block the integrin signaling pathway in vitro and in vivo, we show that there are additional steps that may be targeted therapeutically for inhibiting infection by RNA viruses that traffic to acidic compartments.