Cardiomyocyte-intrinsic SLC25A1 regulates cardiac differentiation and mitochondrial function
Cardiomyocyte-intrinsic SLC25A1 regulates cardiac differentiation and mitochondrial function
Ohanele, C.; Arefeayne, N. A.; Ghazal, N.; Liu, E. H.; Turcanu, N. C.; Horchar, M. J.; Dasgupta, B.; Harbuzariu, A.; Xu, C.; Faundez, V.; Kwong, J. Q.
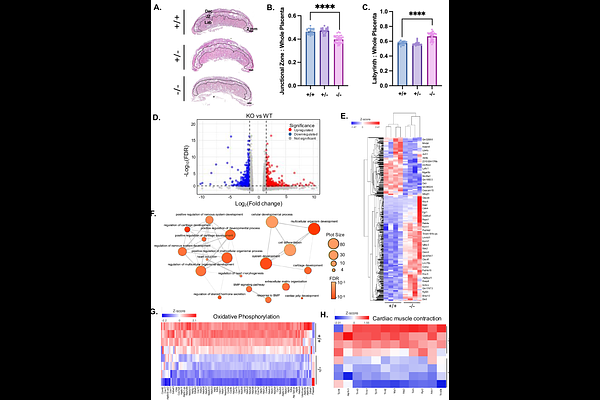
AbstractCardiac morphogenesis is an intricate process that requires a precise coordination between metabolic and structural maturation, but how these processes are linked remain unclear. In previous work, we identified one candidate underlying this connection: the mitochondrial citrate carrier (SLC25A1), a critical regulator of embryonic heart development. Here, using systemic and cardiomyocyte-specific Slc25a1 deletion in mice together with SLC25A1 knockout (KO) human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs), we demonstrate that SLC25A1 functions cell-autonomously within cardiomyocytes to regulate differentiation, mitochondrial maturation, and ventricular morphogenesis. Transcriptomic analysis of SLC25A1-deficient hearts revealed dysregulation of gene programs regulating cardiomyocyte differentiation and mitochondrial function. Consistent with these changes, loss of SLC25A1 in developing cardiomyocytes impaired mitochondrial function and resulted in defective ventricular wall compaction in vivo. Likewise, SLC25A1 KO hiPSC-CMs exhibited defective cardiomyocyte differentiation, disorganized myofibrils, and immature mitochondrial organization and function in vitro. Together, our findings position SLC25A1 as a cardiomyocyte-intrinsic, cell-autonomous regulator that links mitochondrial citrate export to developmental gene programs, revealing a mitochondrial regulatory axis for cardiomyocyte maturation and cardiac morphogenesis that contributes to congenital heart disease.