Crispant analysis in zebrafish as a tool for rapid functional screening of disease-causing genes for bone fragility
Crispant analysis in zebrafish as a tool for rapid functional screening of disease-causing genes for bone fragility
Debaenst, S.; Jarayseh, T.; De Saffel, H.; Bek, J. W.; Boone, M.; Josipovic, I.; Kibleur, P.; Kwon, R. Y.; Coucke, P.; Willaert, A.
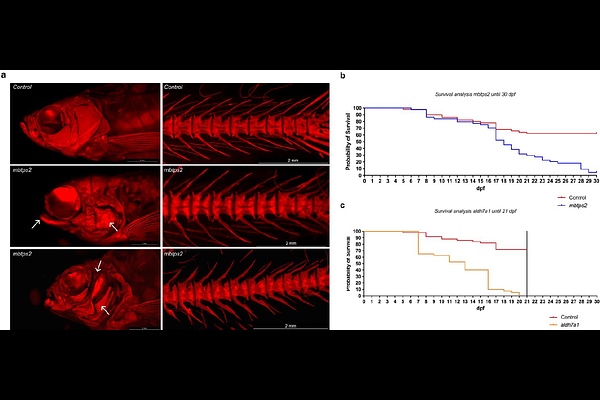
AbstractHeritable Fragile Bone Disorders (FBDs) encompass a spectrum of conditions, from widespread multifactorial to rare monogenic diseases, all characterized by an elevated risk of fractures. The process of validating causative genes and elucidating their pathogenic mechanisms remains a daunting and resource-intensive task. In this study, we evaluated the feasibility of a semi-high throughput zebrafish screening platform for rapid validation and in vivo functional testing and validation of candidate disease-causing genes for a wide range of heritable FBDs. Six genes associated with severe recessive forms of Osteogenesis Imperfecta (OI) and four genes associated with BMD, a key osteoporosis indicator, identified through genome-wide association studies (GWAS) were selected. The crispant screening approach, based on CRISPR/Cas9 technology, was used to phenotype directly in F0 mosaic founder zebrafish. Next-Generation Sequencing (NGS) analysis revealed a mean indel efficiency of 88% across ten different crispants, indicating a high proportion of knock-out alleles and thus resembling stable knock-out models. We applied multiple techniques to evaluate skeletal characteristics at 7, 14 and 90 days post-fertilization (dpf), including microscopy for osteoblast reporter visualization and mineralization by Alizarin Red S staining, and microCT for quantitative skeletal analysis. While larval crispants exhibited variable differences in osteoblast-positive and mineralized surface areas, adult-stage crispants displayed more pronounced and consistent skeletal phenotypes. Notably, all crispants developed malformed neural and haemal arches, with a majority presenting vertebral fractures and fusions, and some showing significant alterations in vertebral bone volume and density. In addition, aldh7a1 and mbtps2 crispants experienced increased mortality due to severe skeletal deformities. RT-qPCR analysis of osteoblast differentiation and bone formation markers at larval stages indicated differential expression of osteogenic markers bglap and col1a1a in a substantial portion of the crispants, hinting at their utility as biomarkers for FBD crispant screening. In summary, our findings demonstrate that crispant screening in zebrafish offers a viable and efficient strategy for the functional assessment of FBD genes. We advocate for a comprehensive approach that integrates various techniques and evaluates distinct skeletal and molecular profiles across different developmental and adult stages. This methodology has the potential to provide new insights into the role of these genes in skeletal biology.