NADP-malic enzyme 1 couples ABA signaling to ROS-auxin patterning to restrict Arabidopsis root growth
NADP-malic enzyme 1 couples ABA signaling to ROS-auxin patterning to restrict Arabidopsis root growth
Fu, Y.; Bouzid, M.; Klamke, M.; Schulze Ising, E.-M.; Sosa, M. M.; Poschmann, G.; Wewer, V.; Metzger, S.; Saigo, M.; Maurino, V. G.
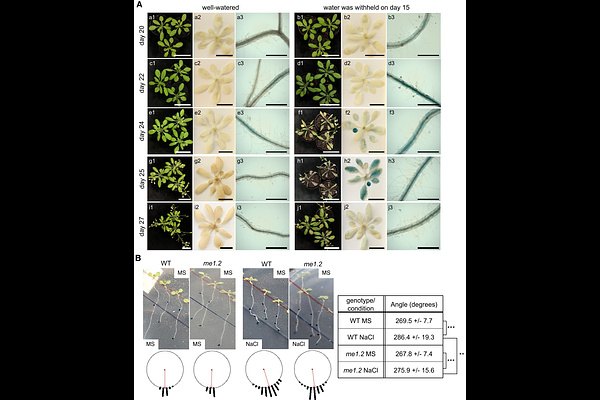
AbstractAbscisic acid (ABA) restricts primary root growth by reshaping reactive oxygen species (ROS) dynamics and hormone signaling at the root apex, yet how cellular reductant supply for redox homeostasis is integrated into this response remains unclear. Here, we show that the cytosolic NADP-dependent malic enzyme 1 (NADP-ME1) is required for full ABA inhibition of Arabidopsis primary root elongation after germination. Three independent me1 loss-of-function mutants retained significant elongation of primary roots under ABA compared with wild type. In wild type, ABA induced an asymmetric auxin response at the root tip, whereas me1 roots failed to establish this auxin asymmetry and instead accumulated superoxide, indicating disrupted ROS balance. Pharmacological perturbation of auxin transport and ethylene biosynthesis/signaling attenuated the mutant phenotype, linking NADP-ME1 function to auxin-ethylene interactions during ABA-regulated growth. NADP-ME1 loss amplifies ABA-dependent transcriptional rewiring, including induction of oxidative stress and depression of growth-associated hormone modules. Co-immunoprecipitation coupled with bimolecular fluorescence complementation identified ascorbate peroxidase 1 (APX1) and major latex protein-like 34 (MLP34) as NADP-ME1 interaction partners, suggesting functional coupling between NADPH production and ROS detoxification. Together, our results support a model in which NADP-ME1 shapes the superoxide/H2O2 balance to control auxin patterning at the root tip and thereby execute ABA-mediated growth inhibition.