Cortical inhibitory parvalbumin interneurons exhibit metabolic specializations coordinated by PGC-1α that are lost in rodents and humans after traumatic brain injury
Cortical inhibitory parvalbumin interneurons exhibit metabolic specializations coordinated by PGC-1α that are lost in rodents and humans after traumatic brain injury
Quinones, S.; Wang, J.; Kim, E.; Bottom-Tanzer, S.; Armbruster, M.; Macedo, J.; Hawkinson, T.; Ribas, R.; Singh, P. K.; Wu, L.; Cantrell, A. R.; Kumar, P.; George, J.; Tai, A.; Whalen, M.; McConnell, M.; Sun, R. C.; Gentry, M. S.; Dulla, C. G.
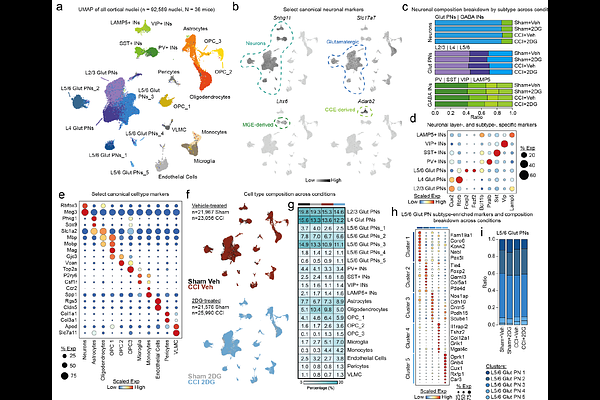
AbstractParvalbumin-positive interneurons (PV-INs) regulate neuronal and circuit activity, and their dysfunction is observed across neurological conditions, including traumatic brain injury (TBI), epilepsy, Alzheimers disease, and schizophrenia. PV-INs are particularly vulnerable to cell loss, potentially due to their increased metabolic demands arising from their uniquely high level of electrical activity, which render them susceptible to metabolic pressure. Here, we use single-nucleus RNA-sequencing (snRNAseq) data from a rodent model of TBI, as well as human TBI data, and demonstrate PV-INs have unique metabolic specializations that are lost after injury and can be rescued by in vivo treatment with the glycolytic inhibitor, 2-deoxyglucose. We generated a novel PV-IN transcriptional identity module comprised primarily of genes encoding specialized ion channels, metabolic enzymes, and synaptic machinery, that identifies heterogenous subsets of injury-associated PV-INs with loss of PV-IN transcriptional identity. We show that changes in metabolic specialization are coupled to changes in transcriptional identity in PV-INs and implicate the PV-IN-enriched transcriptional co-activator, Ppargc1a, as a key driver of PV-IN transcriptional metabolic dysfunction. We also identify a family of long non-coding RNAs enriched in this subset of transcriptionally dysfunctional PV-INs that negatively correlates with PV-IN metabolic specialization. Lastly, we utilize these tools to interrogate a published human TBI snRNAseq data set and find nearly identical changes, underscoring the importance of PV-IN metabolic dysfunction in the pathology of TBI.