Mechanism and cellular function of direct membrane binding by the ESCRT and ERES-associated Ca2+-sensor ALG-2
Mechanism and cellular function of direct membrane binding by the ESCRT and ERES-associated Ca2+-sensor ALG-2
Shukla, S.; Chen, W.; Rao, S.; Yang, S.; Ou, C.; Larsen, K. P.; Hummer, G.; Hanson, P. I.; Hurley, J. H.
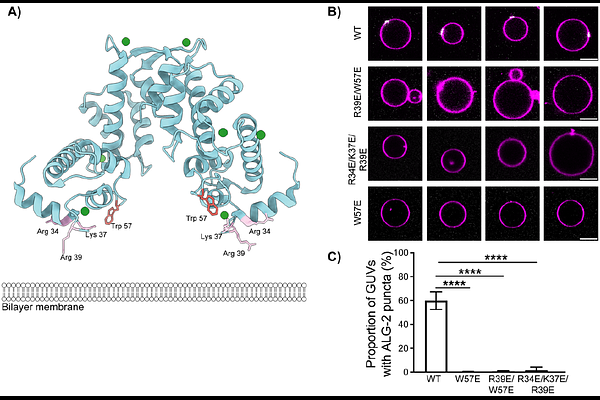
AbstractApoptosis Linked Gene-2 (ALG-2) is a multifunctional intracellular Ca2+ sensor and the archetypal member of the penta-EF hand protein family. ALG-2 functions in the repair of damage to both the plasma and lysosome membranes and in COPII-dependent budding at endoplasmic reticulum exit sites (ERES). In the presence of Ca2+, ALG-2 binds to ESCRT-I and ALIX in membrane repair and to SEC31A at ERES. ALG-2 also binds directly to acidic membranes in the presence of Ca2+ by a combination of electrostatic and hydrophobic interactions. By combining GUV-based experiments and molecular dynamics simulations, we show that charge-reversed mutants of ALG-2 at these locations disrupt membrane recruitment. ALG-2 membrane binding mutants have reduced or abrogated ERES localization in response to Thapsigargin-induced Ca2+ release but still localize to lysosomes following lysosomal Ca2+ release. In vitro reconstitution shows that the ALG-2 membrane-binding defect can be rescued by binding to ESCRT-I. These data thus reveal the nature of direct Ca2+-dependent membrane binding and its interplay with Ca2+-dependent protein binding in the cellular functions of ALG-2.