Filament-resolved simulations reproduce self-organization of lamellipodia and filopodia
Filament-resolved simulations reproduce self-organization of lamellipodia and filopodia
Fukui, M.; Kondo, Y.; Saito, N.; Naoki, H.
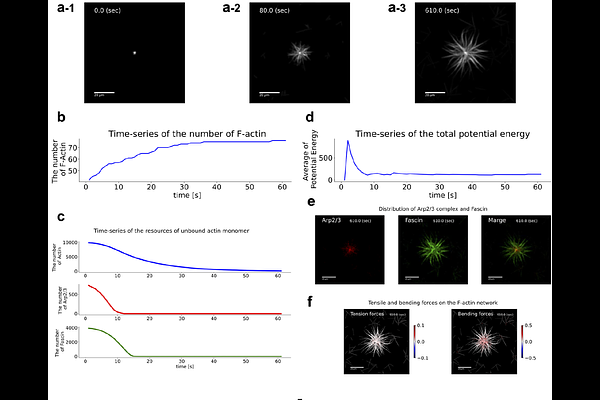
AbstractThe dynamic assembly of actin filaments underlies diverse cellular morphologies such as lamellipodia, filopodia, and reticulated networks. However, how filament-scale interactions among actin-binding proteins produce distinct actin architectures remains unclear. We developed a filament-resolved computational model of actin self-organization regulated by the Arp2/3 complex and fascin. Individual F-actin filaments are represented as elastic chains, and their stochastic polymerization, Arp2/3-mediated branching, and fascin-mediated crosslinking and bundling are explicitly modeled. The simulations reproduce three actin architectures observed in minimal reconstitution experiments, including lamellipodia-like branched networks, filopodia-like bundled protrusions, and reticulated meshworks, as a function of Arp2/3 and fascin concentrations. We quantify these regimes using actin density, orientational order, and spikiness, which robustly separate the three morphologies across conditions. To connect filament organization to shape change, we further couple the actin network to membrane deformation using a phase-field formulation. This coupling shows how localized remodeling concentrates load to drive pseudopodial protrusions, whereas highly branched networks distribute stresses and stabilize rounded shapes. The model links molecular interactions to emergent architecture and cell-scale morphodynamics.