From gHBfix to NBfix: Reweighting-Driven Refinement of Hydrogen-Bond Interactions in RNA Force Fields
From gHBfix to NBfix: Reweighting-Driven Refinement of Hydrogen-Bond Interactions in RNA Force Fields
Mlynsky, V.; Kuehrova, P.; Bussi, G.; Otyepka, M.; Sponer, J.; Banas, P.
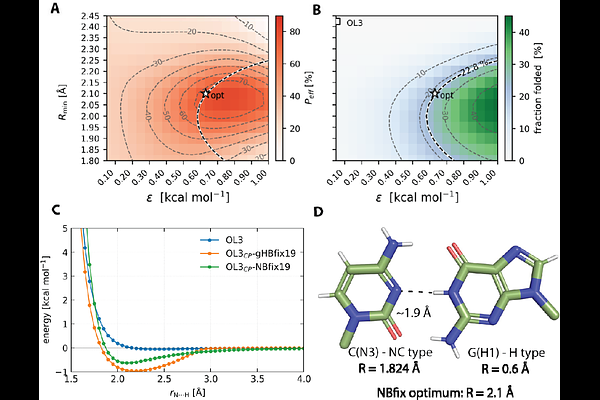
AbstractUnderstanding RNA structural dynamics is essential for elucidating its biological functions, and molecular dynamics (MD) simulations provide an important atomistic complement to experimental approaches. However, the predictive power of MD is fundamentally limited by the accuracy of the underlying empirical Force Fields (FFs), particularly in capturing the delicate balance of non-bonded interactions. Here, we present a systematic reparameterization strategy that replaces the external gHBfix19 hydrogen-bond (H-bond) correction potential with an equivalent set of NBfix Lennard-Jones modifications within a state-of-the-art RNA FF. Using a quantitatively converged temperature replica-exchange MD ensemble of the GAGA tetraloop, we employed a reweighting-based optimization protocol to derive NBfix parameters that reproduce the thermodynamic effects of the original gHBfix19 terms. Sequential optimization of individual gHBfix19 components proved essential to ensure stable and transferable parameter refinement. The resulting fully reformulated NBfix-based variant, termed OL3CP--NBfix19, was validated on a representative set of RNA motifs, including tetranucleotides, A-form duplexes, and tetraloops. Across all tested systems, its performance is comparable to that of the reference gHBfix19 FF. By embedding the H-bond corrections directly into the standard non-bonded framework, the NBfix formulation eliminates external biasing potentials, simplifies practical deployment, and reduces computational overhead. Beyond this specific reparameterization, our results demonstrate a practical workflow for translating targeted H-bond corrections into native FF terms for efficient biomolecular simulations.