Establishment and comprehensive characterization of a novel dark-reared zebrafish model for myopia studies
Establishment and comprehensive characterization of a novel dark-reared zebrafish model for myopia studies
Xie, J.; Goodbourn, P. T.; Bui, B. V.; Jusuf, P. R.
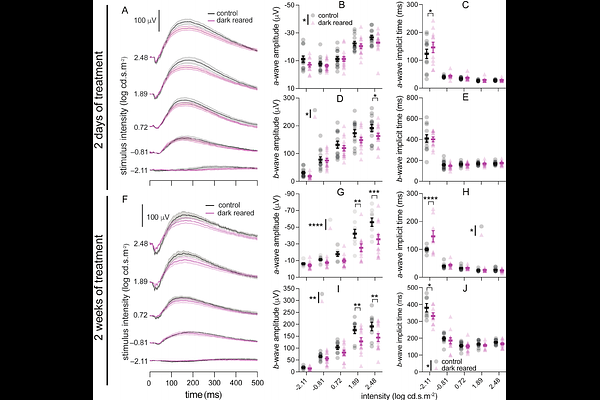
AbstractPurpose. Myopia is predicted to impact approximately 5 billion people by 2050, necessitating mechanistic understanding of its development. Myopia results from dysregulated genetic mechanisms of emmetropization, caused by over-exposure to aberrant visual environments; however, these genetic mechanisms remain unclear. Recent human genome-wide association studies have identified a range of novel myopia-risk genes. To facilitate large-scale in vivo mechanistic examination of gene-environment interactions, this study aims to establish a myopia model platform that allows efficient environmental and genetic manipulations. Methods. We established an environmental zebrafish myopia model by dark-rearing. Ocular biometrics including relative ocular refraction were quantified using optical coherence tomography images. Spatial vision was assessed using optomotor response (OMR). Retinal function was analyzed via electroretinography (ERG). Myopia-associated molecular contents or distributions were examined using RT-qPCR or immunohistochemistry. Results. Our model produces robust phenotypic changes, showing myopia after 2 weeks of dark-rearing, which were recoverable within 2 weeks after returning animals to normal lighting. 2-week dark-reared zebrafish have reduced spatial-frequency tuning function. ERG showed reduced photoreceptor and bipolar cell function (a- and b-waves) after only 2 days of dark-rearing, which worsened after 2 weeks of dark-rearing. We also found dark-rearing-induced changes to expression of myopia-risk genes, including egr1, vegfaa, vegfab, rbp3, gjd2a and gjd2b, inner retinal distribution of EFEMP1, TIMP2 and MMP2, as well as transiently reduced PSD95 density in the inner plexiform layer. Conclusions. Coupled with the gene editing tools available for zebrafish, our environmental myopia model provides an excellent platform for large-scale investigation of gene-environment interactions in myopia development.