Vagal Signaling Decline in Age-related Macular Degeneration Drives Spleen-Dependent Retinal Inflammation
Vagal Signaling Decline in Age-related Macular Degeneration Drives Spleen-Dependent Retinal Inflammation
Ronning, K. E.; Roubeix, C.; Nous, C.; Augustin, S.; Blond, F.; Argyriou, F.; Molbech, C. R.; Veres, K.; Mehlsen, J.; Bak, M.; Gautier, E. L.; Sorensen, H. T.; Guillonneau, X.; Delarasse, C.; Sorensen, T. L.; Sennlaub, F.
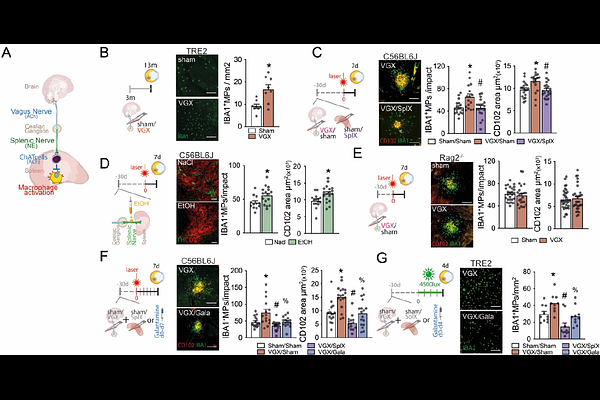
AbstractUnder relaxed physiological conditions, vagal innervation via the splenic nerve restrains the release of inflammatory cytokines from splenic macrophages and helps preserve systemic homeostasis, constituting the efferent cholinergic anti-inflammatory arm of the inflammatory reflex. Our analysis of Danish National Patient Registry data revealed that vagotomy for peptic ulcer disease, particularly truncal vagotomy, markedly increased the risk of developing age-related macular degeneration (AMD). Consistently, experimental truncal vagotomy or splenic denervation in mice exacerbated laser- and light-induced subretinal inflammation, both models of late AMD. The pro-inflammatory effects of vagotomy were abolished by concurrent splenectomy or pharmacologically augmenting acetylcholine signaling. Mechanistically, vagotomy activated a pro-inflammatory transcriptional program in splenic monocytes while suppressing tissue-retention genes Cxcr4 and Fn1, leading to enhanced monocyte egress from the spleen and increased infiltration into the injured retina. In the laser-injured retina, single-cell RNA sequencing (scRNAseq) of mononuclear phagocytes revealed a broad set of vagotomy-induced transcripts in infiltrating monocytes and activated microglia. Remarkably, more than one-third of these were normalized by splenectomy. Together, these findings identify a vagus nerve-spleen-retina axis that connects stress and vagal tone to AMD pathogenesis.