Parkinson's disease linked LRRK2 G2019S drives oxidative nuclear DNA damage and PARP1 hyperactive signaling
Parkinson's disease linked LRRK2 G2019S drives oxidative nuclear DNA damage and PARP1 hyperactive signaling
Liu, J.; Gonzalez-Hunt, C. P.; Richbourg, T.; Barraza, I.; Chen, C.; Montes, C.; Ma, L.; Cao, R.; Hanumaihgari, V.; Gassman, N. R.; Fouquerel, E.; Sanders, L. H.
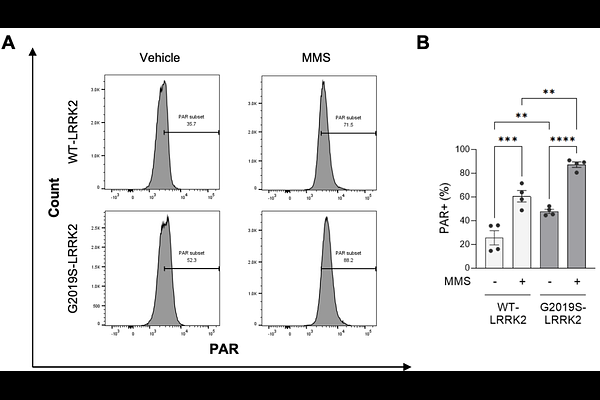
AbstractLRRK2 mutations are the most common cause of autosomal-dominant Parkinson's disease (PD), with G2019S linked to both familial and sporadic PD. Although LRRK2-mediated mitochondrial DNA damage is implicated in PD, the contribution of nuclear DNA damage is less understood. Using CRISPR/Cas9-generated LRRK2G2019S/G2019S knock-in cells, we discovered increased sensitivity to oxidative and alkylating DNA-damaging agents compared to wild-type, consistent with compromised tolerance/repair of lesions processed by base excision repair (BER). The oxRADD assay revealed elevated endogenous oxidative nuclear base damage in LRRK2 mutant cells. Concomitantly, PARP1-dependent poly(ADP-ribose) (PAR) levels were markedly increased, with chromatin enrichment of PARP1 and BER factors (XRCC1, DNA ligase III) only in LRRK2G2019S/G2019S cells, indicating BER initiation, without successful resolution. LRRK2G2019S/G2019S cells displayed synthetic lethality with PARP-trapping inhibitors (olaparib) but tolerated PARP1 knockdown, suggesting cytotoxicity from stabilized PARP-DNA complexes rather than loss of catalytic activity. The SOD/catalase mimetic EUK-134 abrogated LRRK2 G2019S-dependent PAR accumulation, whereas the mitochondrial complex I inhibitor rotenone exacerbated PAR levels, linking reactive oxygen species (ROS) to BER dysfunction and PARP1 hyperactivation. Overall, we have identified a ROS-dependent PARP1 hyperactivation pathway that underlies LRRK2 G2019S-associated cellular vulnerability.