A structure-selective endonuclease drives uniparental mitochondrial DNA inheritance
A structure-selective endonuclease drives uniparental mitochondrial DNA inheritance
Shimomura, M.; Yun, H. Y.; Zuzarte, P. C.; Simpson, J. T.; Wyatt, H. D. M.; Hurd, T. R.
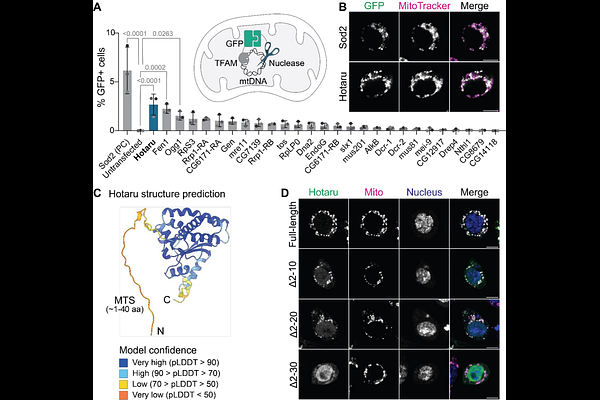
AbstractMaternal inheritance of mitochondrial DNA (mtDNA) is a near-universal feature of eukaryotes, yet the mechanisms that ensure this by preventing paternal mtDNA inheritance have remained unclear. In both Drosophila and humans, mtDNA is actively eliminated from sperm during spermatogenesis, producing mature sperm whose mitochondria lack their genomes. Here we identify Hotaru, a previously uncharacterized, testis-specific GIY-YIG endonuclease, as a central player in this process. We find that Hotaru is expressed in elongated spermatids, localizes to the mitochondrial matrix, and is required for paternal mtDNA elimination. In hotaru mutants, sperm retain mtDNA at levels comparable to those present before the elimination process. Genetic and biochemical analyses show that Hotaru selectively recognizes and cleaves cruciform DNA structures within the mtDNA control region. Together, these findings identify a dedicated nuclease that enforces mitochondrial genome elimination in the animal male germline and reveal that an unexpected structural feature of mtDNA serves as the molecular determinant of its destruction. By recognizing DNA structure rather than specific sequence motifs, this mechanism is inherently robust to the high mutation rate of mitochondrial genomes.