Revealing the Biophysics of Lamina-Associated Domain Formation by Integrating Theoretical Modeling and High-Resolution Imaging
Revealing the Biophysics of Lamina-Associated Domain Formation by Integrating Theoretical Modeling and High-Resolution Imaging
Dhankhar, M.; Guo, Z.; Kant, A.; Basir, R.; Joshi, R.; Heo, S. C.; Mauck, R. L.; Lakadamyali, M.; Shenoy, V. B.
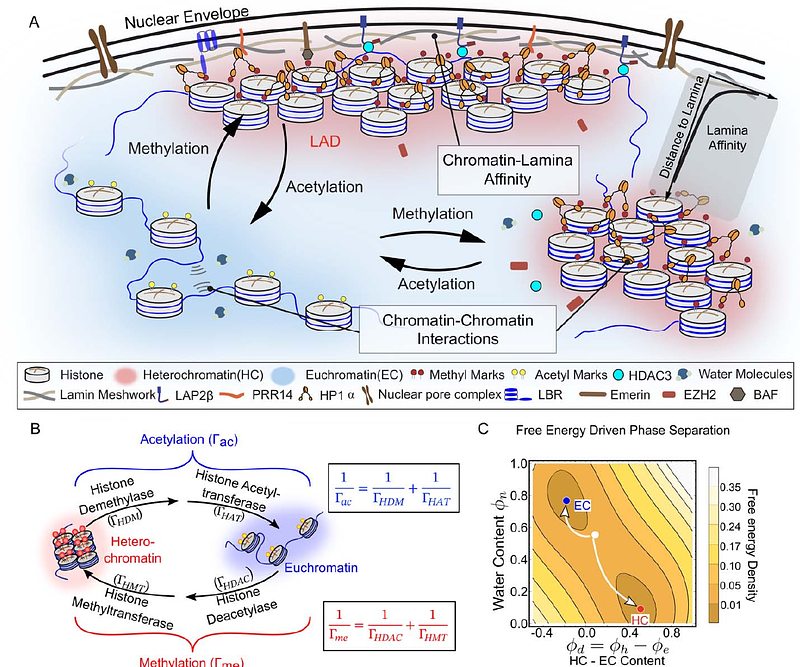
AbstractThe interactions between chromatin and the nuclear lamina orchestrate cell type-specific gene activity by forming lamina-associated domains (LADs) which preserve cellular characteristics through gene repression. However, unlike the interactions between chromatin segments, the strength of chromatin-lamina interactions and their dependence on cellular environment are not well understood. Here, we develop a theory to predict the size and shape of peripheral heterochromatin domains by considering the energetics of chromatin-chromatin interactions, the affinity between chromatin and the nuclear lamina and the kinetics of methylation and acetylation in human mesenchymal stem cells (hMSCs). Through the analysis of super-resolution images of peripheral heterochromatin domains using this theoretical framework, we determine the nuclear lamina-wide distribution of chromatin-lamina affinities. We find that the extracted affinity is highly spatially heterogeneous and shows a bimodal distribution, indicating regions along the lamina with strong chromatin binding and those exhibiting vanishing chromatin affinity interspersed with some regions exhibiting a relatively diminished chromatin interactions, in line with the presence of structures such as nuclear pores. Exploring the role of environmental cues on peripheral chromatin, we find that LAD thickness increases when hMSCs are cultured on a softer substrate, in correlation with contractility-dependent translocation of histone deacetylase 3 (HDAC3) from the cytosol to the nucleus. In soft microenvironments, chromatin becomes sequestered at the nuclear lamina, likely due to the interactions of HDAC3 with the chromatin anchoring protein LAP2 beta increasing chromatin-lamina affinity, as well as elevated levels of the intranuclear histone methylation. Our findings are further corroborated by pharmacological interventions that inhibit contractility, as well as by manipulating methylation levels using epigenetic drugs. Notably, in the context of tendinosis, a chronic condition characterized by collagen degeneration, we observed a similar increase in the thickness of peripheral chromatin akin to that of cells cultured on soft substrates consistent with theoretical predictions. Our findings underscore the pivotal role of the microenvironment in shaping genome organization and highlight its relevance in pathological conditions.


