Autophagy impairment by ATG4B deficiency reduces experimental hypersensitivity pneumonitis severity
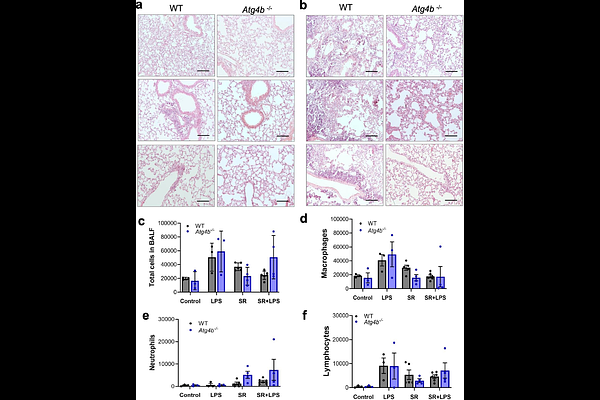
Autophagy impairment by ATG4B deficiency reduces experimental hypersensitivity pneumonitis severity
Cabrera, S.; Sanchez-Barajas, A.; Gaxiola, M.; Garcia-Vicente, A.; Selman, M.; Pardo, A.
AbstractAutophagy has been implicated in several lung diseases, either protecting tissues or driving pathology. Hypersensitivity pneumonitis (HP) is a complex inflammatory lung disease, and autophagy is heavily involved in regulating inflammation. The role of autophagy in HP remains unclear. The aim of our study was to understand the role of autophagy in HP pathogenesis. GFP-LC3 transgenic mice were exposed intranasally to Saccharopolyspora rectivirgula (SR) to induce HP and follow autophagy activation in the lung. Then, we take advantage of our Atg4b-deficient mouse model to assess how autophagy disruption impacts lung inflammation in response to SR antigen challenge. Increased autophagy activation was observed in epithelial and inflammatory cells after SR antigen exposure in GFP-LC3 transgenic lungs. GFP-LC3 puncta colocalized with ATG4B and ATG5 in epithelial and inflammatory cells after antigen exposure. Autophagy impairment limits the inflammatory response after SR antigen exposure in the lungs from the Atg4b-deficient mice when compared to WT mice. To evaluate whether lipopolysaccharide (LPS) exacerbates the inflammatory response in the Atg4b-deficient, a SR+LPS combined treatment was developed and we discovered that LPS aggravates the SR-induced HP in WT but not in Atg4b-deficient mice. Reduced HP severity in Atg4b-deficient mice was associated with decreased expression of NFkB, CCL1, CCL25, CXCL1, TNFR1, IL-13, and IL-17A, diminished CD4+ T cell recruitment and expansion, reduced M2-like macrophages, and decreased granuloma and iBALT development. Our findings highlight autophagy as a critical driver in HP pathogenesis and as a potetial target for novel theraphy development.