Generative Deep Learning and Molecular Dynamics Reveal Design Principles for Amyloid-Like Antimicrobial Peptides
Generative Deep Learning and Molecular Dynamics Reveal Design Principles for Amyloid-Like Antimicrobial Peptides
Prasad, A. K.; Awatade, V.; Patel, M. K.; Plisson, F.; Martin, L.; Panwar, A. S.
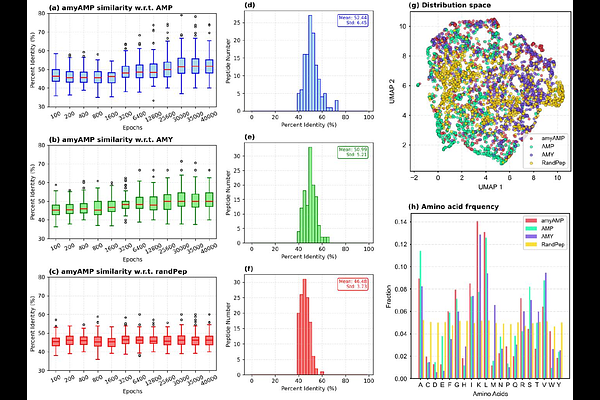
AbstractAntimicrobial peptides (AMPs) are emerging as promising alternatives to conventional antibiotics, and growing evidence indicates a fundamental link between antimicrobial activity and amyloidlike self-assembly. Many AMPs are known to form amyloid-like fibrils, while several amyloidogenic peptides exhibit intrinsic antimicrobial properties, suggesting shared underlying physicochemical determinants such as amphipathicity, {beta}-sheet propensity, and charge distribution. However, the rational design of peptides that simultaneously encode these dual functionalities remains a significant challenge. Here, we present amyAMP, a generative deep-learning framework based on a Wasserstein generative adversarial network with gradient penalty (WGAN-GP), designed to learn and generate peptides with integrated antimicrobial and amyloidogenic properties. Trained on curated datasets of antimicrobial and amyloid-forming peptides, amyAMP captures the latent sequence-property relationships governing dual functionality. Statistical and latent-space analyses demonstrate that the generated peptides closely overlap with biologically relevant peptide space while remaining distinct from random sequences, indicating successful learning of key biochemical features. To validate functional behavior, we performed extensive coarse-grained molecular dynamics simulations to probe membrane interaction, peptide selfassembly, and membrane disruption. The simulations reveal rapid membrane adsorption, stable amphipathic insertion, and strong peptide-peptide aggregation. Notably, cooperative clustering of peptides on membrane surfaces induces membrane thinning and curvature perturbations, highlighting a mechanistic coupling between aggregation and antimicrobial activity. Collectively, these results establish that amyAMP effectively captures the shared physicochemical principles underlying antimicrobial action and amyloid-like self-assembly. This work provides a generalizable framework for the AI-guided design of multifunctional peptides to advance the development of next-generation therapeutics targeting antimicrobial resistance.