A human synovial tendon-on-a-chip models key features of peritendinous adhesions and offers a new approach methodology for testing anti-fibrotic drugs
A human synovial tendon-on-a-chip models key features of peritendinous adhesions and offers a new approach methodology for testing anti-fibrotic drugs
Linares, I.; Creveling, A.; Osman, A.; Grandwetter, N.; Miller, B. M.; McGrath, J. L.; Awad, H. A.
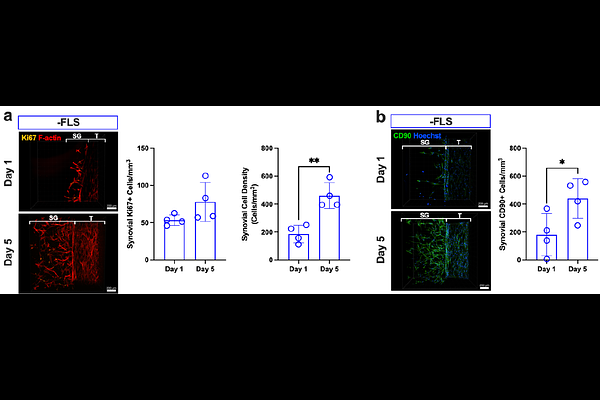
AbstractPeritendinous adhesions are a debilitating complication of tendon injury characterized by excessive matrix deposition and chronic inflammation. Due to limitations of current preclinical models, the underlying mechanisms of adhesion pathogenesis remain poorly defined, and there are no approved drugs to prevent or resolve adhesions. Here, we develop a human synovial tendon-on-a-chip (synToC) that integrates synovial fibroblasts, tendon-resident fibroblasts, immune cells, and vascular endothelium to reconstruct the intrasynovial tendon microenvironment. We show that synovial fibroblast activation promoted tendon contraction and inflammatory cytokine secretion dominated by IL-6, leading to monocyte infiltration and formation of fibronectin- and collagen III-rich matrix bridges between tendon and synovial compartments resembling nascent peritendinous adhesions. These phenotypes emerged even in the absence of exogenous TGF-{beta}1, indicating that synovial fibroblast-mediated crosstalk is sufficient to initiate adhesion-like pathology. Importantly, pharmacological inhibition of the IL-6/JAK/STAT pathway suppressed synovial activation, blunted inflammatory cytokine signaling, and attenuated fibrotic matrix deposition and interfacial adhesion formation. These findings establish the synToC as a human-relevant new approach methodology (NAM) to interrogate the multicellular drivers of tendon adhesions and to accelerate the development of anti-fibrotic therapies.