Intrinsic electrostatics of ATP synthase modulate the proton motive force across species
Intrinsic electrostatics of ATP synthase modulate the proton motive force across species
K. Matar, I.; Fahimi, P.; Vigneau, J.-N.; Matta, C. F.
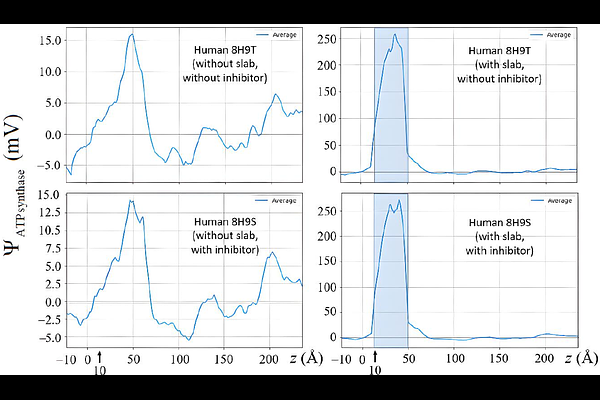
AbstractATP synthase is conventionally viewed as a passive consumer of the proton-motive force (pmf). Here, analysis of 178 crystallographic and cryo-EM structures from 17 species reveals that the enzyme itself carries an intrinsic electrostatic potential (ESP) that contributes directly to the electrical component of the pmf. Poisson-Boltzmann calculations show that approximately half of the species exhibit a constructive enzyme-linked voltage of magnitudes up to ~ 20 mV, whereas other species - including Homo sapiens - exhibit an opposing contribution of comparable magnitude. These enzyme-linked voltages correspond to shifts of up to ~ 2 kJ/mol per proton, representing a measurable fraction of the electrical work available for ATP synthesis. The intrinsic electrostatic term represents an electrical contribution, hitherto overlooked, to the electric part of the pmf. This term does not alter the global electrochemical gradient imposed by the membrane; rather, it modifies the effective voltage drop experienced locally by the Fo motor, thereby influencing how proton free energy is partitioned between ATP production and dissipation. In humans, the opposing polarity of the enzyme's ESP reduces the effective driving voltage at Fo by up to ~ 10%, potentially shifting the balance between ATP yield and heat release under tightly coupled conditions. These findings identify ATP synthase's electrostatics as a previously unrecognized, sequence-dependent modifier of oxidative phosphorylation that provides a structural basis for variability in the thermodynamic efficiency of ATP production and the mechanical efficiency of the rotary motor across species and potentially among individuals.