Coordinated immune dysregulation in Juvenile Dermatomyositis revealed by single-cell genomics
Coordinated immune dysregulation in Juvenile Dermatomyositis revealed by single-cell genomics
Rabadam, G.; Wibrand, C.; Flynn, E.; Hartoularos, G. C.; Sun, Y.; Ye, C. J.; Kim, S.; Gartner, Z. J.; Sirota, M.; Neely, J.
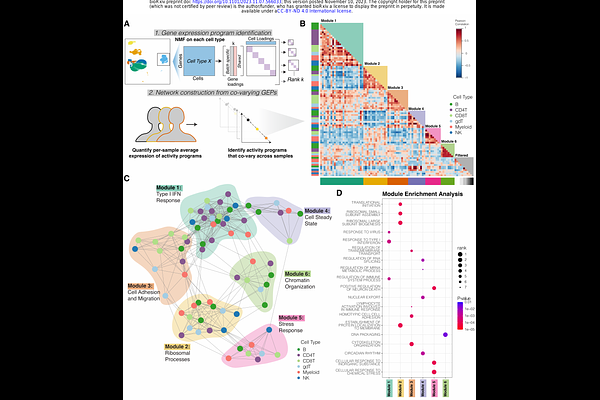
AbstractJuvenile Dermatomyositis (JDM) is one of several childhood-onset autoimmune disorders characterized by a type I interferon response and autoantibodies. Treatment options are limited due to incomplete understanding of how the disease emerges from dysregulated cell states across the immune system. We therefore investigated the blood of JDM patients at different stages of disease activity using single-cell transcriptomics paired with surface protein expression. By immunophenotyping peripheral blood mononuclear cells, we observed skewing of the B cell compartment towards an immature naive state as a hallmark of JDM. Furthermore, we find that these changes in B cells are paralleled by signatures of Th2-mediated inflammation. Additionally, our work identified SIGLEC-1 expression in monocytes as a composite measure of heterogeneous type I interferon activity in disease. We applied network analysis to reveal that hyperactivation of the type I interferon response in all immune populations is coordinated with dysfunctional protein processing and regulation of cell death programming. This analysis separated the ubiquitously expressed type I interferon response into a central hub and revealed previously masked cell states. Together, these findings reveal the coordinated immune dysregulation underpinning JDM and provide novel insight into strategies for restoring balance in immune function.