A fast and accurate calculation method for light induced isomerization of retinal proteins in real time
A fast and accurate calculation method for light induced isomerization of retinal proteins in real time
Althoff, P.; Labudda, K.; Hoeweler, U.; Luebben, M.; Gerwert, K.; Koetting, C.; Rudack, T.
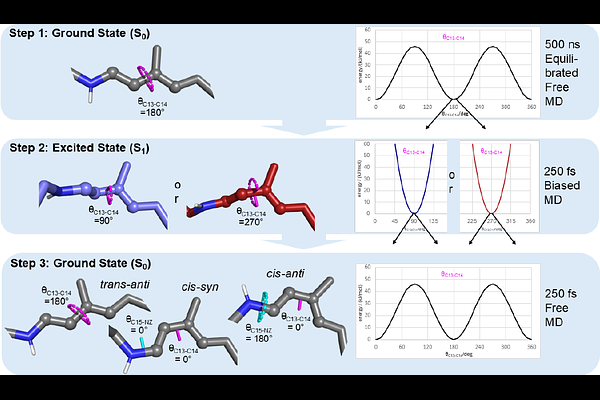
AbstractRetinal is a chromophore covalently bound to various photoreceptors. Its photo-induced isomerization triggers a series of structural changes named photocycle, leading to diverse biological functions. Despite tremendous advances in structural biology and artificial intelligence-driven structure prediction, it remains challenging to analyze all photocyclic intermediates. Here, we present an optimized computational approach to calculate RSBH+ isomerization and its induced structural changes based on a classical molecular mechanics approach using quantum mechanically improved retinal force field parameters. Isomerization is induced by an excited state restraint which is subsequently relaxed to allow the return to the electronic ground state. We applied this approach to the key protein of optogenetics, Channelrhodopsin-2 from Chlamydomonas reinhardtii (CrChR2). Besides the reformation of the all-trans/CN-anti ground state, we observed the production of a mixture of two isomeric states 13-cis/CN-anti and 13-cis/CN-syn. These findings agree with the previously found branched photocycle model based on experimental data. Our calculations show an asymmetric potential energy landscape of the excited state leading to a corresponding isomerization state distribution. Unlike earlier publications, our procedure describes the retinal photoisomerization on the natural timescale of 500 fs. As our newly derived retinal force field parameter set precisely relies on quantum biological knowledge, it assists to improve the refinement of experimental structure biological data. Our readily customizable strategy provides mechanistic insights at high spatio-temporal resolution, which permits accurate structural predictions of early photocycle intermediates. These insights will stimulate the rational design of optogenetic tools thus providing improved diagnostic and therapeutic approaches for neuronal and other diseases.