The genome assembly of Sphaerospora molnari provides novel insights into the rapid evolution and diversification of unique lineage-specific gene groups in myxozoan parasites
The genome assembly of Sphaerospora molnari provides novel insights into the rapid evolution and diversification of unique lineage-specific gene groups in myxozoan parasites
Kosakyan, A.; Wisniewska, M.; Riera-Ferrer, E.; Alama-Bermejo, G.; Fiala, I.; Karlikova, A.; Dirks, R. P.; Kolisko, M.; Holzer, A.
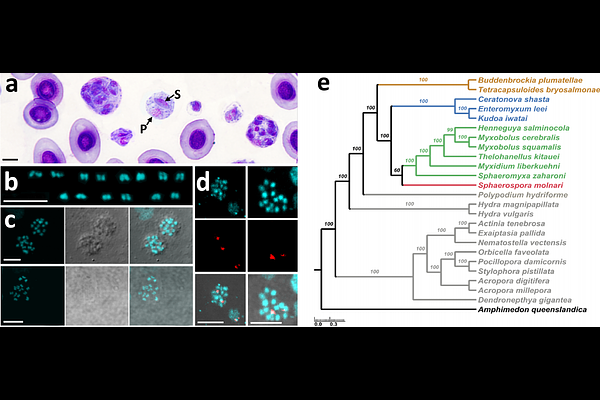
AbstractBackground Myxozoans are ancient cnidarian parasites with highly derived genomes characterized by an extremely accelerated rate of nucleotide substitution, abundant orphan and lineage-specific genes without further characterization. Genome data are limited to two out of the four main evolutionary lineages, assemblies are highly fragmented and often show significant levels of host contamination. Results We present a near chromosome-scale myxozoan genome, based on Oxford Nanopore-reads of Sphaerospora molnari, a member of the previously uncharacterized group of blood-feeding myxozoans, thereby addressing a key gap in phylogeny of the subphylum. The haploid genome assembly spans 40.17 Mb in 40 contigs, harbors 14,957 genes and shows the smallest mitogenome of myxozoans (14,015 bp). Gene gain/gene loss analyses showed that myxozoans have small ancestral gene repertoires and show highly lineage-specific genome compositions. Using comparative analyses focusing on identifying unique but diversified gene compartments in S. molnari, we discovered taxonomically restricted protist genes related to red blood cell attachment (Plasmodium ETRAMPs) and surface protein variation (Plasmodium variant surface antigen families RIFIN and STEVOR, as well as Metamonada variant-specific surface proteins, VSPs), raising questions about their origins and evolution. A genomic trait shared between several myxozoans is the significant expansion of DNA transposable elements belonging to the mutator-like elements (MULEs), and while the simple copy-paste mechanism of these transposases may suggest frequent uncontrolled mutation, we demonstrate domestication of MULEs into transcription factors. Analyses of the gene fragments of chimeric MULEs (pack-MULES) of S. molnari show that these coincide with highly diversified gene groups in this lineage, including alien genes, suggesting MULEs as a driving force for gene evolution in myxozoans. Conclusions Parasitic lifestyle shifts drive exceptionally rapid genome evolution in myxozoans, primarily through nonadaptive mutation and gene transfer via MULEs, with adaptive refinement through MULE domestication and selection. In S. molnari, these processes underpin unique erythrocyte exploitation and immune evasion strategies essential for survival in the bloodstream of the host.