The Dynamic Nature of Genetic Risk for Schizophrenia Within Genes Regulated by FOXP1 During Neurodevelopment
The Dynamic Nature of Genetic Risk for Schizophrenia Within Genes Regulated by FOXP1 During Neurodevelopment
Ali, D.; Donohoe, G.; Morris, D. W.
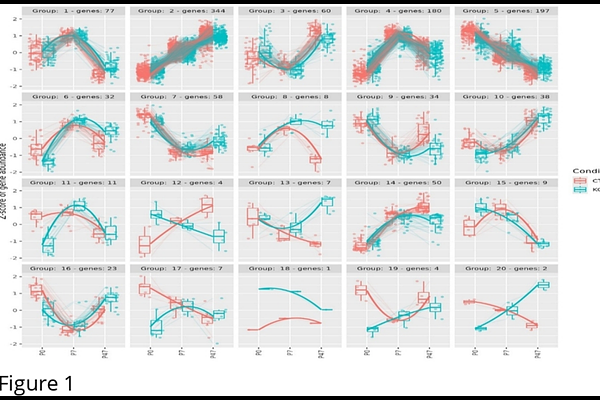
AbstractFOXP1 (Forkhead-box protein P1) is a crucial transcription factor in neural development and is associated with schizophrenia (SCZ). FOXP1-regulated genes may contribute to genetic risk of SCZ and this may vary across different stages of neurodevelopment. We analyzed transcriptomic data from mouse and human models of FOXP1 loss-of-function across prenatal and postnatal developmental stages, including neural stem cells from embryonic mice (E14.5) and human brain organoids (equivalent to second trimester), and cortical tissues from different mouse postnatal stages P0, P7, and P47. P0 in mice corresponds to the third trimester in humans, while P7 and P47 represent early childhood and adolescence, respectively. The effect of FOXP1 disruption on gene expression in cortical tissues/cells was assayed using RNA-seq, including time-course and pairwise gene expression analysis. Linkage disequilibrium score regression assessed if FOXP1-regulated genes were enriched for SCZ heritability. Gene-set enrichment analysis investigated if FOXP1-regulated genes were enriched for SCZ-associated genes reported as differentially expressed in single cortical cell studies. SynGO analysis mapped FOXP1-regulated genes to synaptic locations and functions. FOXP1-regulated genes are enriched for SCZ heritability, with significant results for E14.5, P7 and P47 but not P0. The P7 gene-set showed the strongest enrichment for SCZ-associated genes from single cortical cell studies. FOXP1-regulated genes at both P7 and P47 were involved in multiple synaptic functions and were mainly enriched within glutamatergic excitatory neurons, with P47 also showing enrichment within GABAergic inhibitory neurons across regions of the postnatal cortex. Prenatal FOXP1-regulated genes were enriched in progenitor cells and also mapped to the synapse. Genetic risk for SCZ within FOXP1-regulated genes follows a dynamic trajectory across developmental stages, showing stronger effects at timepoints that map to early childhood, followed by adolescence and second trimester.