Metabolic adaptation to maternal hyperglycemia via ACLY-dependent acetyl-CoA production drives epigenetic remodeling and dysregulated placental development
Metabolic adaptation to maternal hyperglycemia via ACLY-dependent acetyl-CoA production drives epigenetic remodeling and dysregulated placental development
Liu, M.; Jin, K.; QI, S.; Chen, D.; Han, Y.; Xu, W.; Wen, C.; Wen, H.; Liu, Y.; He, B.; Lin, X.
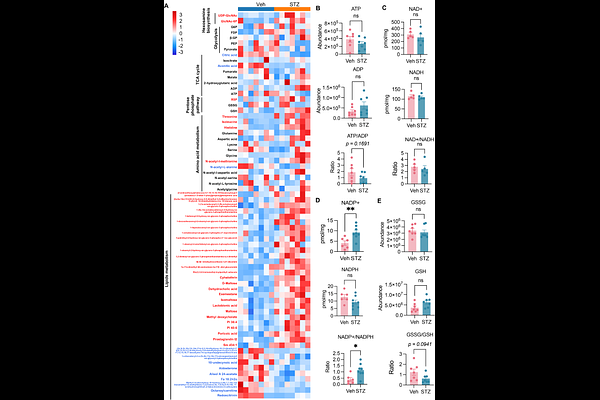
AbstractGestational diabetes mellitus (GDM) is a common metabolic complication of pregnancy that is paradoxically associated with both fetal overgrowth and fetal growth restriction (FGR). While maternal hyperglycemia is widely presumed to drive macrosomia through excessive nutrient supply, the mechanisms underlying FGR remain poorly understood. Here, using a mouse model that recapitulates the small-for-gestational-age (SGA) phenotype observed in human GDM pregnancies, we identify placental underdevelopment as a principal driver of FGR. Despite systemic nutrient abundance, hyperglycemic placentas exhibit reduced mass and an increased fetal-to-placental weight ratio, indicative of placental insufficiency. Mechanistically, maternal hyperglycemia induces anabolic metabolic rewiring while suppressing oxidative phosphorylation (OXPHOS), accompanied by upregulation and nuclear redistribution of ATP-citrate lyase (ACLY). ACLY converts glucose-derived carbon into acetyl-CoA in the cytosol and nucleus, thereby coupling glycolytic flux to lipid and hexosamine biosynthesis as well as to global histone hyperacetylation. This hyperacetylation-associated epigenetic reprogramming activates metabolic, innate immune, and inflammatory gene programs while repressing pro-proliferative and anti-apoptotic pathways. Consequently, placental growth is compromised despite nutrient excess. Importantly, activation of the ACLY-acetyl-CoA axis and global histone hyperacetylation is consistently observed in human GDM placentas across diverse birth outcomes, suggesting a conserved metabolic-epigenetic adaptation to maternal hyperglycemia. Together, these findings identify ACLY-dependent acetyl-CoA production as a central metabolic node linking maternal hyperglycemia to chromatin remodeling and placental development control, thereby reshaping fetal growth trajectories.