Multiple Sclerosis Drug Fingolimod Exhibits Antibacterial Activity through Bacterial Membrane Permeabilization
Multiple Sclerosis Drug Fingolimod Exhibits Antibacterial Activity through Bacterial Membrane Permeabilization
Syam, A.; Rees, B.; Cuervo, S.; Xue, F.; Sodt, A.; Nestorovich, E. M.; Rostovtseva, T.; Bezrukov, S. M.; Choy, J.
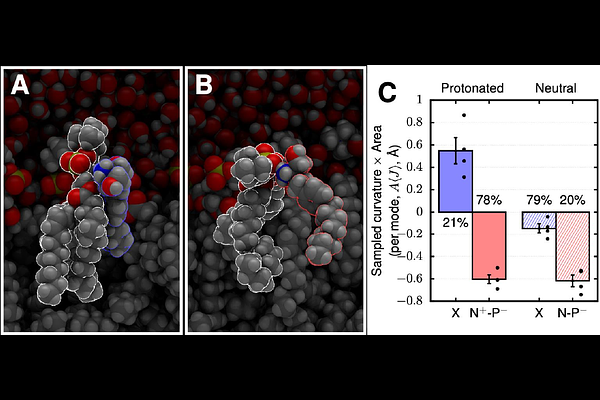
AbstractAlthough receptor-mediated mechanisms account for the therapeutic action of numerous FDA-approved drugs, emerging evidence suggests that many of these therapeutics have off-target antimicrobial activities. One example is fingolimod, an immunomodulator used to treat multiple sclerosis, that has been reported to have antimicrobial effects associated with membrane permeabilization. Yet, the molecular mechanism by which fingolimod alters bacterial membranes remains unknown. As a cationic amphiphilic drug (CAD), fingolimod is comprised of both hydrophobic and positively charged regions that can enable membrane interactions. We show that fingolimod compromises membrane integrity in E. coli and P. aeruginosa, contributing to its antimicrobial activity. To determine how fingolimod compromises membrane integrity, we used planar lipid bilayer electrophysiology with phospholipid compositions mimicking E. coli membranes. Using gramicidin A channels as molecular biosensors, we show that fingolimod alters both mechanical properties and surface charge of lipid bilayers at concentrations relevant to its antimicrobial effects. At concentrations exceeding 4 {micro}M, fingolimod directly permeabilizes lipid bilayers, revealed by conductance measurements and Bilayer Overtone Analysis. Molecular dynamics simulations correlate fingolimods preference for pore-favoring curvature with its strong interactions with lipids and trans-leaflet translocation. These findings establish a molecular mechanism for fingolimod's off-target activity and provide a starting point for understanding how some CAD structures can drive membrane-specific effects that compromise bacterial physiology.