A family of bacterial actin homologues forms a 3-stranded tubular structure
A family of bacterial actin homologues forms a 3-stranded tubular structure
Bergeron, J.; Kollman, J. M.
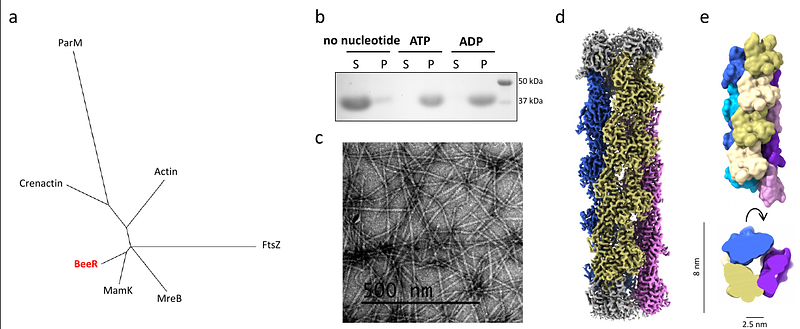
AbstractThe cytoskeleton plays a critical role in the organization and movement of cells. In Eukaryotes, actin filaments polymerize into a highly conserved double-stranded linear filamentous structure in the presence of ATP, and disassemble upon ATP hydrolysis. Bacteria also possess actin-like proteins, that drive fundamental cellular function, including cell division, shape maintenance, and DNA segregation. Like eukaryotic actin, bacterial actins assemble dynamic on ATP binding. In general, longitudinal interactions between bacterial actin protomers along each strand are conserved with eukaryotic actin, but variation in interactions between strands gives rise to striking diversity of filament architectures. Here, we report a family of bacterial actins of unknown function, conserved amongst the Verrucomicrobiota phylum, which assembles into a unique tubular structure in the presence of nucleotides. A cryo-EM structure of the filaments reveals it is composed of three strands, unlike other described bacterial actin structures. This architecture provides new insights into the organization of actin-like filaments, and has implications for understanding the diversity and evolution of the bacterial cytoskeleton.