Label-Free 4D Holotomography with Depth-Adaptive Segmentation for Quantitative Analysis of Lipid Droplet Dynamics in Hepatic Organoids
Label-Free 4D Holotomography with Depth-Adaptive Segmentation for Quantitative Analysis of Lipid Droplet Dynamics in Hepatic Organoids
cho, j.; lee, h.; oh, c.; park, j.; park, s.; koo, b.-k.; Park, Y.
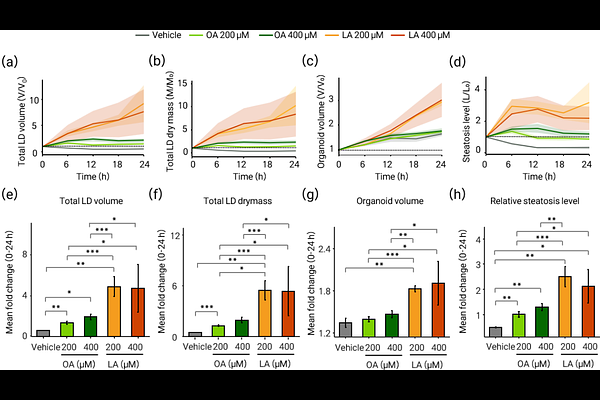
AbstractStatement of Discovery: We developed a label-free, longitudinal 3D holotomography framework with depth-adaptive lipid droplet segmentation that quantifies single-droplet dynamics in living mouse hepatic organoids. Using this platform, we found that oleic acid and linoleic acid induce lipid droplet (LD) accumulation via distinct strategies-oleic acid via droplet enlargement and linoleic acid via sustained increases in droplet number-while palmitic acid rapidly compromises organoid integrity. Significance: Quantifying LD remodeling in 3D liver organoids is often limited to endpoint staining or phototoxic live fluorescence imaging, thereby obscuring droplet-level kinetics. Aim: We aimed to develop a label-free method to track LD dynamics in living hepatic organoids under different fatty-acid loads. Approach: Time-lapse 3D refractive-index tomograms were acquired using holotomography and analyzed with a depth-adaptive, multi-threshold segmentation pipeline to quantify LD number, volume, sphericity, and refractive-index-derived lipid concentration and dry mass at single-droplet resolution. Results: Oleic acid and linoleic acid induced LD accumulation while preserving organoid integrity, whereas palmitic acid triggered rapid structural collapse. Despite increases in total LD burden under both oleic acid and linoleic acid, droplet-level dynamics diverged: oleic acid produced volume-dominated accumulation via enlargement of fewer LDs and increased size heterogeneity, whereas linoleic acid produced number-dominated accumulation via sustained increases in LD number, yielding a more uniform population of small droplets. Conclusions: Label-free holotomography with depth-adaptive analysis enables non-invasive, longitudinal, and multi-scale quantification of LD dynamics in intact organoids and reveals fatty-acid-dependent temporal modes of lipid storage.