UBA1 Mitigates Myocardial Ischemia/Reperfusion Injury by Attenuating Endoplasmic Reticulum-Mitochondria Contacts via Pdzd8 ubiquitination
UBA1 Mitigates Myocardial Ischemia/Reperfusion Injury by Attenuating Endoplasmic Reticulum-Mitochondria Contacts via Pdzd8 ubiquitination
Xu, L.-L.; Li, P.-B.; Jiang, W.; Du, J.; Li, H.-H.
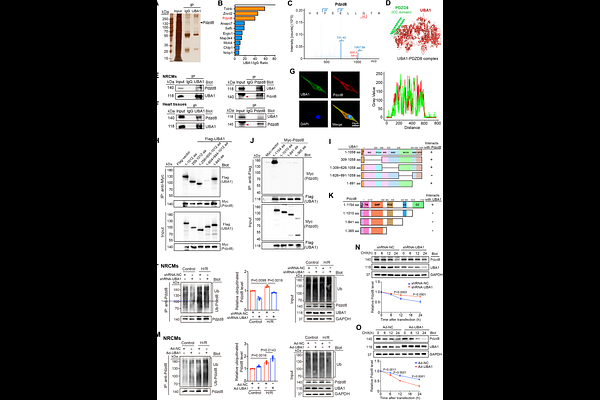
AbstractBACKGROUND: Myocardial ischemia/reperfusion injury (I/RI) represents a serious clinical complication in patients after acute myocardial infarction. Ubiquitin-activating enzyme 1 (UBA1) catalyzes the initial step of ubiquitination and plays a fundamental role in regulating protein homeostasis and related diseases. This study aims to elucidate the functional contribution of UBA1 to the pathogenesis of myocardial I/RI and to uncover its underlying mechanisms. METHODS: Single-cell RNA sequencing was employed to characterize UBA1 expression in human ischemic heart tissues. Myocardial I/R injury was examined in myocardial-specific UBA1 knockout (UBA1cko) mice, UBA1-overexpressing mice (rAAV9-UBA1), and corresponding controls. Neonatal rat cardiomyocytes underwent hypoxia/reoxygenation in vitro. Cardiac function and infarction were evaluated by echocardiography and pathological staining. Protein?protein interactions were analyzed via immunoprecipitation combined with mass spectrometry. The endoplasmic reticulum?mitochondrial contact sites (ERMCSs) and mitochondrial ultrastructure were evaluated through transmission electron microscopy and confocal imaging. RESULTS: UBA1 expression was significantly downregulated in human and murine ischemic myocardium, especially in cardiomyocytes. UBA1cko mice exhibited aggravated I/RI with greater infarct size, impaired function, apoptosis, elevated intracellular Ca2+ levels, mitochondrial dysfunction, and ER stress, whereas UBA1 overexpression conferred cardioprotective effects. Mechanistically, UBA1 directly bound to and ubiquitinated Pdzd8, a key ERMCS-tethering protein, thereby promoting its degradation, which inhibited ERMCS formation and improved mitochondrial dysfunction and ER stress. Moreover, knockdown of Pdzd8 via rAAV9-siRNA effectively mitigated UBA1 knockout-induced myocardial damage. Additionally, administration of auranofin (AF), a U.S. Food and Drug Administration-approved drug for treating rheumatoid arthritis, markedly alleviated myocardial I/RI via activating UBA1 in vivo and in vitro.