The atypical filament assembly underpins the inflammasome-independent functions of IFI16.
The atypical filament assembly underpins the inflammasome-independent functions of IFI16.
Garg, A.; Niedzialkowska, E.; Zhou, J.; Moh, J.; Egelman, E.; Sohn, J.
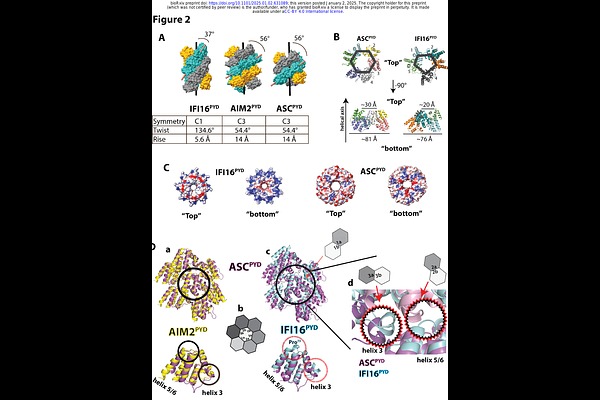
AbstractInflammasomes trigger cell death upon sensing various intracellular maladies. These supra-structures relay upstream signals by sequentially assembling architecturally congruent filaments via their pyrin domains (PYDs). Interferon Inducible Protein 16 (IFI16) is an innate immune sensor that detects dysregulated nucleic acids. Once presumed as an inflammasome receptor due to its PYD, the role of IFI16 has been much more appreciated in other innate immune pathways such as regulating interferon production and viral replication restriction. Here, a cryo-EM structure of the filament assembled by the PYD of IFI16(PYD) shows a helical architecture distinct from inflammasome PYD filaments. In silico Rosetta interaction energy calculations suggest that the helical architecture of the IFI16PYD filament is incompatible with those assembled by the central inflammasome adaptor ASC and its interacting partners. Cellular experiments further support that IFI16 fails to interact with ASC. Together, we provide the structural basis for the inflammasome-independent functions of IFI16.