Activation of PPARγ redirects fibro-adipogenic progenitors to replace ectopic bone with fat in models of fibrodysplasia ossificans progressiva and trauma-induced heterotopic ossification
Activation of PPARγ redirects fibro-adipogenic progenitors to replace ectopic bone with fat in models of fibrodysplasia ossificans progressiva and trauma-induced heterotopic ossification
Koirala, P.; Chen, Z.; Hanumantharao, S. N.; Siegel, A. E.; Liu, C.; Williams, Z.; Sekhon, H.; Maridas, D.; Mishina, Y.; Rosen, V.; Agarwal, S.
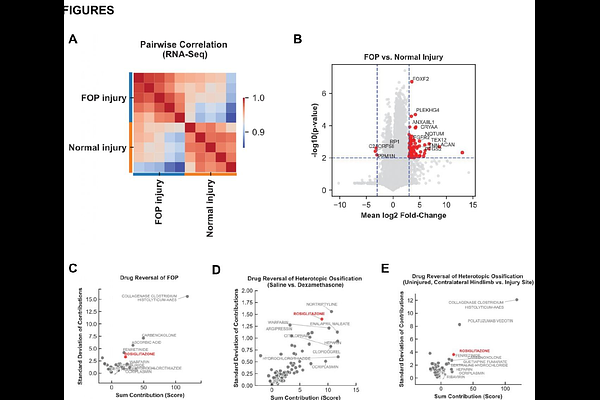
AbstractThe pathologic, osteogenic differentiation of fibroadipogenic progenitor cells (FAPs) is the primary recognized contributor to ectopic bone formation in fibrodysplasia ossificans progressiva (FOP) and trauma-induced heterotopic ossification (HO). Both conditions are characterized by up-regulated BMP signaling; the former by a gene mutation rendering the BMP receptor ACVR1 susceptible to activation by inflammatory ligands (Activin A), and the latter by up-regulated presence of BMP2 ligand in the setting of unmutated BMP receptor. We performed an unbiased assessment of FDA-approved therapies which would optimally target the transcriptional aberrations observed in developing FOP and HO lesions based on publicly-available datasets. This analysis uncovered rosiglitazone, a peroxisome proliferator-activated receptor gamma (PPAR{gamma}) agonist as the highest scoring therapeutic option across three data sets for both conditions. Rosiglitazone treatment eliminated ectopic bone lesions in a mouse model of FOP, and replaced these lesions with ectopic adipose tissue; similarly, systemic and local rosiglitazone treatment eliminated ectopic bone lesions in a mouse model of trauma-induced HO and replaced these lesions with ectopic adipose tissue. Our findings were corroborated by a single case report from 2010 showing positive results with rosiglitazone in a non-diabetic patient with FOP, with no subsequent studies. Overall, our findings suggest that a previously FDA-approved therapeutic is likely to be a successful therapeutic agent for both FOP and trauma-induced HO, both conditions for which current therapeutic options remain inadequate.