Common γ-chain cytokines induce an epigenomically plastic precursor-like KIT+ ILC2 state linked to immune disease susceptibility
Common γ-chain cytokines induce an epigenomically plastic precursor-like KIT+ ILC2 state linked to immune disease susceptibility
Olsthoorn, S. E. M.; Onrust-Van Schoonhoven, A.; de Bruijn, M. J. W.; van Nimwegen, M.; van Beek, G.; de Koning, W.; Trap, L.; van der Ploeg, E. K.; Sanders, M. A.; Surace, L.; Di Santo, J. P.; Hendriks, R. W.; Stadhouders, R.
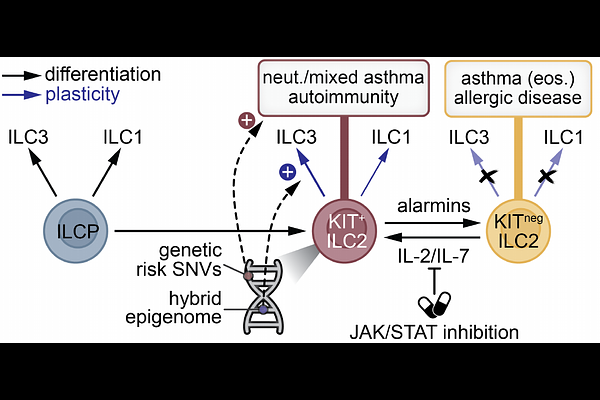
AbstractBackground: Group 2 innate lymphoid cells (ILC2s) are key effector cells of type-2 immunity. A subset of ILC2s expresses KIT (CD117), which display increased phenotypic plasticity and were previously linked to severe asthma and psoriasis. However, the molecular mechanisms promoting a KIT+ ILC2 state remain poorly understood. Objective: Define the molecular basis for the enhanced plasticity of KIT+ ILC2s and identify signals that induce this phenotype, including links with immune disease susceptibility. Methods: We combine bulk as well as single-cell transcriptome (RNA-seq) and epigenome (ATAC-seq) with in vitro culture assays using primary human KIT+ or KITneg ILC2s and multipotent ILC precursors (ILCPs). Epigenomic data were integrated with genetic risk variants for major human immune diseases. Results: Multi-omics analyses revealed that KIT+ ILC2s maintain a unique hybrid character marked by expression and open chromatin of genes linked to both ILCP and ILC2 biology. KIT+ ILC2s showed extensive epigenomic priming at gene loci related to naive lymphocyte biology, tissue homing, and ILC3 effector functions, including IL17 and IL23R - explaining why KIT+ ILC2s are poised to adopt an ILC3-like phenotype. Genetic risk variants for asthma and autoimmunity are enriched in the poised epigenome of KIT+ ILC2s. Common {gamma}-chain cytokines IL-2 and IL-7 induced a KIT+ phenotype in KITneg ILC2s through STAT5 activation. Conclusions: Our study defines KIT+ ILC2s as a developmentally immature state carrying a precursor-like epigenome that promotes phenotypic plasticity and is linked to immune disease susceptibility. Importantly, we identify STAT5-mediated cytokine signals as candidates for therapeutic targeting of KIT+ ILC2s.