Aging modifies microstructure and material properties of mineralized cartilage and subchondral bone in the murine knee
Aging modifies microstructure and material properties of mineralized cartilage and subchondral bone in the murine knee
Müller, L.; Blouin, S.; Pedrinazzi, E.; van Lenthe, G. H.; Hego, A.; Weinkamer, R.; Hartmann, M. A.; Ruffoni, D.
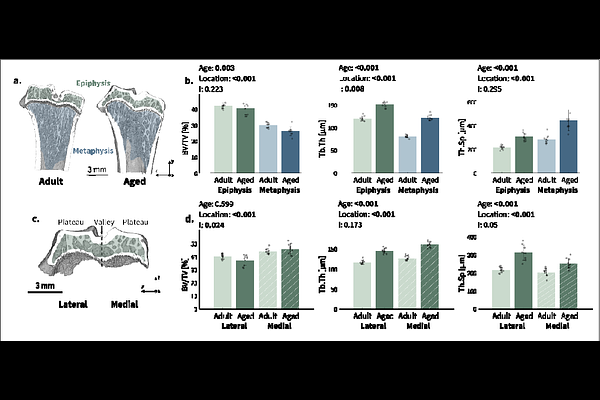
AbstractThe osteochondral junction is a specialized region ensuring the biomechanical and biological integration of the unmineralized articular cartilage with the subchondral bone through an intermediate layer of mineralized cartilage. This location is of clinical relevance, being the target of osteoarthritis. While aging is considered a risk factor for osteoarthritis, the interplay between microstructural and material changes during aging and predisposing to joint degeneration is not fully clear. This is especially true for mineralized cartilage, which remains understudied despite its critical role in load transfer from unmineralized articular cartilage to bone. We investigate age-related alterations of mineralized cartilage and subchondral bone in rat tibiae of adult and aged animals using a multimodal, high-resolution, correlative analysis. Our approach includes micro-computed tomography to measure microstructural features, second harmonic generation imaging to visualize collagen organization, quantitative backscattered electron imaging to map local mineral content, and nanoindentation to obtain mechanical properties. Mineralized cartilage and subchondral bone exhibited distinct age-related modifications. At the architectural level, the subchondral plate thickened and the trabecular network became coarser, those changes being different from those observed in the metaphysis. At the tissue level, mineralized cartilage was less mineralized than bone but exhibits a greater relative increase of mineral content with age, underlying differences in mineralization. A central observation is that aging led to an abrupt transition in mineral content and mechanical properties across the interface between unmineralized and mineralized cartilage, with a conceivable impact on stress localization. Overall, these changes may alter load transfer and contribute to age-related joint degeneration.