Dimerisation and twist reversal of the Lewy fold in α-synuclein mutants with Parkinson's disease and dementia
Dimerisation and twist reversal of the Lewy fold in α-synuclein mutants with Parkinson's disease and dementia
Zhang, H.; Murzin, A. G.; Macdonald, J. A.; Hinton, T. V.; Peak-Chew, S.; Franco, C.; Cullinane, P. W.; Warner, T.; Okuzumi, A.; Real, R.; Nishioka, K.; Taniguchi, D.; Kaneda, D.; Morris, H.; Houlden, H.; Ghetti, B.; Jaunmuktane, Z.; Yang, Y.; Scheres, S.; Goedert, M.
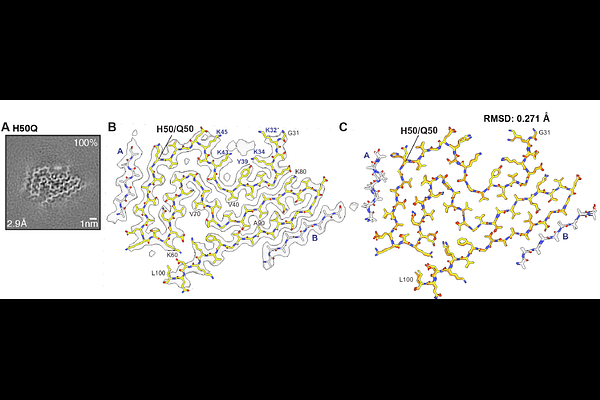
AbstractDominantly inherited missense and gene dosage mutations in SNCA, the -synuclein gene, cause familial forms of Parkinson's disease and dementia. Here we report the structures of -synuclein filaments from the brains of such individuals. Pathogenic mutations A53T and G51D in SNCA give rise to singlets and doublets of the Lewy fold with a left-handed helical twist in the absence of a peptide-like density for island A. By contrast, filaments from the non-pathogenic variant H50Q consist of singlets of the right-handed Lewy fold with a density for island A, like filaments of wild-type -synuclein. The structures of filaments from homozygous mice transgenic for human mutant A53T -synuclein (line M83) are unlike those from human brains. They are more similar to the multiple system atrophy folds than to the Lewy fold of Parkinson's disease, Parkinson's disease dementia and dementia with Lewy bodies