Sub-chronic peripheral CB1R inhibition enhances cognitive performance and induces hippocampal synaptic plasticity changes in naive mice
Sub-chronic peripheral CB1R inhibition enhances cognitive performance and induces hippocampal synaptic plasticity changes in naive mice
Bergada-Martinez, A.; de los Reyes-Ramirez, L.; Martinez-Torres, S.; Martinez-Gallego, I.; Maldonado, R.; Rodriguez-Moreno, A.; Ozaita, A.
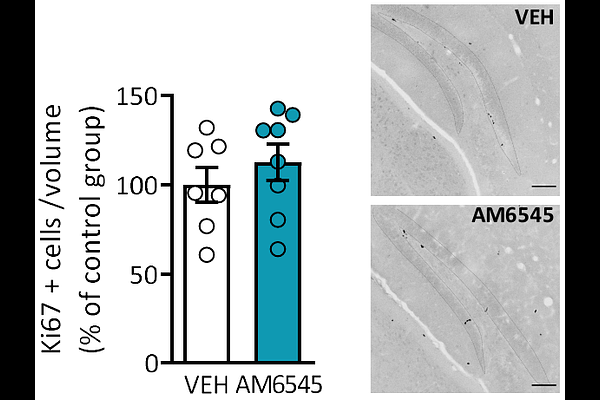
AbstractThe peripheral contribution to brain function and cognitive performance is far from understood. Cannabinoid type-1 receptor (CB1R), a well-recognized player in cognitive function, is classically pictured in the central nervous system to have such a role. Our group previously demonstrated a novel mechanism where the acute peripheral CB1R inhibition in mice promotes low arousal memory persistence. Here, to elucidate the associated synaptic modifications involved, we evaluated the cognitive outcomes as well as cellular and molecular consequences of a sub-chronic treatment with the peripherally-restricted CB1R antagonist AM6545. Sub-chronic AM6545 resulted in enhanced memory persistence in the novel object-recognition memory test in both males and females and also improving emotional memory. In addition, executive function was facilitated after repeated AM6545 administration further strengthening the nootropic properties of peripheral CB1R inhibition. Transcriptional analysis of hippocampal synaptoneurosomes from treated mice revealed changes in the transcript expression of subunit 1 of N-methyl-D-aspartate (NMDA) receptor isoforms. Notably, AM6545 treatment occluded long-term potentiation in hippocampal synapses while enhancing input-output relation. These changes in synaptic plasticity were accompanied by an increase in the hippocampal expression of Bdnf and Ngf neurotrophic factors. Our results suggest that repeated peripheral CB1R inhibition contributes to the modulation of memory persistence, executive function, and hippocampal synaptic plasticity in mice, further indicating that peripheral CB1R could act as targets for a novel class of nootropic compounds.