TurboID-based proteomic profiling reveals proxitome of the IRT1 metal transporter and new insight into metal uptake regulation in plants
TurboID-based proteomic profiling reveals proxitome of the IRT1 metal transporter and new insight into metal uptake regulation in plants
Pellegrin, L.; Fanara, S.; Fabre, B.; Pichereaux, C.; Cotelle, V.; Vert, G.; Neveu, J.
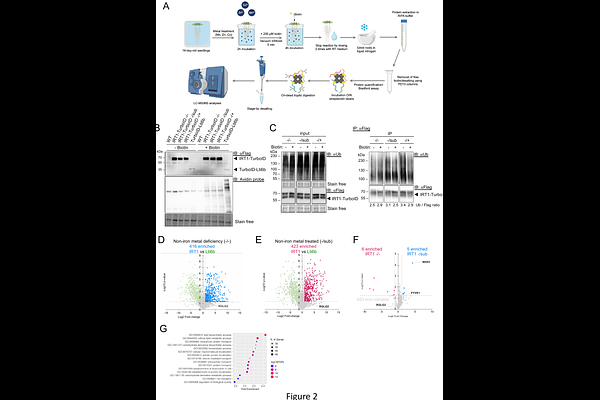
AbstractIRT1 is the major root iron transporter responsible for broad spectrum metal absorption in Arabidopsis root epidermal cells. Non-iron metal substrates of IRT1 were recently shown to regulate IRT1 cell surface levels by endocytosis and vacuolar degradation. TurboID-based proximity labeling was recently developed to detect protein:protein interactions and thus shed light on the intricate regulation of proteins in living cells. Although TurboID-based proximity labeling technology has been successfully established in mammals, its application in plant systems remains limited and inexistent for highly hydrophobic multispann transmembrane proteins. Here, we established TurboID for proximity labeling of IRT1 and identified 494 IRT1-specific proximal proteins, including the previously reported FYVE1 IRT1 interacting protein. To showcase the biological relevance of identified IRT1 proximal proteins, we characterized further the NHX5 Na+(K+)/H+ antiporter and the RGLG2 E3 ubiquitin ligase. We validated both IRT1 proximal proteins as IRT1 partners using several orthogonal assays. We also highlight the contribution of NHX-type antiporters and RGLG-type E3 ligases in plants responses to non-iron metal nutrition and IRT1 endocytosis. Overall, our work showcases the power of TurboID to identify new interacting proteins for plant transporters, expanding the application of this technology to proteins notoriously difficult to work with.