SARS-CoV-2 PLpro Drives Epithelial Barrier Disruption Across Drosophila and Mammalian Epithelia
SARS-CoV-2 PLpro Drives Epithelial Barrier Disruption Across Drosophila and Mammalian Epithelia
Ahmad, Q. T.; Banerjee, S.; Anerao, I. M.; Tripathi, J.; Parihar, S. S.; Bajpai, A.; Rizvi, M. S.; SINHA, P.
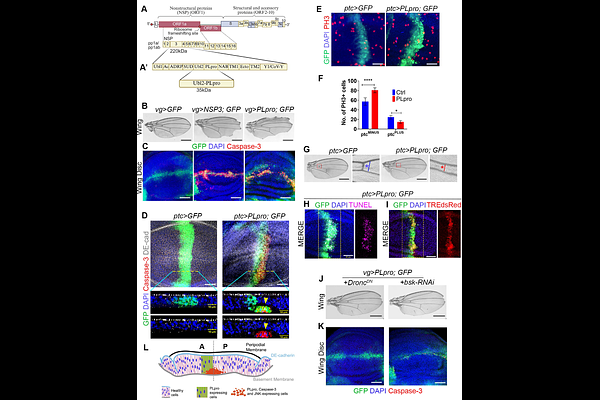
AbstractDisruption of epithelial homeostasis and barrier integrity, compounded by maladaptive inflammation, is central to SARS-CoV-2 pathogenesis. Prior reductionist studies relying on mammalian cell lines, which lack the capacity to form architecturally faithful epithelium in vitro, were ill-suited to identify the viral proteins that proximally underpin these epithelial perturbations. We therefore screened all SARS-CoV-2 NSPs and ORFs in the Drosophila larval imaginal disc, an intact epithelium whose conserved junctional architecture and stress signaling circuits closely recapitulate those of human epithelium. This systemic filter identified the papain-like protease (PLpro) of NSP3 as a primary driver of junctional disruption. In imaginal disc epithelium, PLpro expression induced elevated oxidative stress (ROS) and hyperactivation of Akt, JNK, and JAK-STAT pathways, a self-amplifying pathological network that acts as a 'cog-in-the-wheel' to derail tissue homeostasis. PLpro further induced organ-specific junctional perturbations and dysregulation of stress signaling in the larval tracheal system and the adult midgut, extending its epithelial-disrupting role across three morphologically distinct organs. Targeted genetic suppression of this network restored integrity, establishing these pathways as causal drivers. Critically, PLpro expression in mammalian MDCK epithelium recapitulated these defects, revealing cross-species conservation. These findings redefine PLpro as a 'dual-threat' pathogen, not merely an immune-evasion factor, but a direct 'architect of injury' and a previously unrecognized determinant of COVID-19 pathogenesis.