Fronto-temporal structural alterations in congenital aphantasia
Fronto-temporal structural alterations in congenital aphantasia
Takamura, Y.; Delsanti, R.; Cohen, L.; Bartolomeo, P.; liu, J.
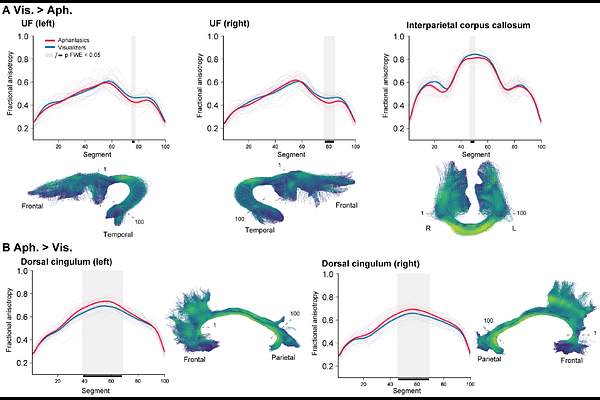
AbstractCongenital aphantasia is characterized by a lifelong absence of voluntary visual imagery despite preserved visual knowledge, offering a natural model for dissociating sensory representation from conscious imagery experience. Functional imaging evidence suggests that this dissociation may arise from altered top-down interactions between higher-order control systems and high-level visual cortex, but its structural correlates remain unknown. Here, using diffusion and structural MRI in 18 individuals with congenital aphantasia and 18 matched visualisers, we tested two competing accounts of aphantasia: one predicting structural differences in visual pathways, the other predicting differences in higher-order associative networks. Across complementary analyses of white-matter tract microstructure, functional-ROI tractography, graph-theoretic network organization, and cortical morphometry, aphantasia was associated with selective differences in bilateral uncinate fasciculus, posterior interparietal callosal fibers, bilateral dorsal cingulum, left anterior insula, and anterior prefrontal and medial temporal cortex. By contrast, we found no reliable group differences in early visual cortex, major visual tracts, or the direct structural connections of the core imagery network. Congenital aphantasia therefore exhibits a selective structural phenotype centered on fronto-temporal and cingulate systems, sparing the principal visual pathways. These findings suggest that higher-order systems supporting integration, regulation, and conscious access -rather than visual representations themselves -constitute the primary structural substrate of congenital aphantasia.